Content Menu
● Why Reconstitute Lidocaine Powder?
● Materials Needed for Reconstitution
● Step-by-Step Guide to Reconstituting Lidocaine Powder
>> Step 1: Preparation and Safety
>> Step 2: Choosing the Correct Diluent
>> Step 3: Calculating Diluent Volume
>> Step 4: Mixing Diluent into the Powder
>> Step 5: Dissolving the Powder Completely
>> Step 6: Drawing up the Solution
>> Step 7: Administration and Disposal
● Important Tips and Precautions
● Advanced Considerations for Clinical and Manufacturing Use
>> Stability and Sterility Monitoring
● Frequently Asked Questions (FAQs)
>> 1. How long can reconstituted lidocaine solution be stored before use?
>> 2. Can I use normal saline or sterile water as a diluent for lidocaine powder?
>> 3. Is it safe to add adrenaline to lidocaine during reconstitution?
>> 4. What concentration of lidocaine is commonly used for numbing injections?
>> 5. What is the maximum safe dose for lidocaine injection?
Lidocaine is one of the most widely used local anesthetics in modern medicine. It plays a vital role in numbing tissues to provide pain relief during minor surgical procedures, dental work, or the insertion of medical devices. When supplied in powder form, lidocaine requires careful reconstitution into an injectable solution to ensure both safety and efficacy. This article provides an in-depth guide on how to properly reconstitute lidocaine powder for numbing injections, explaining the necessary materials, step-by-step procedures, safety considerations, and practical tips.
Our company specializes in biotechnology, pharmaceutical health, and medical devices, providing reliable OEM services to global brand owners, wholesalers, and manufacturers. This comprehensive guide supports healthcare professionals and manufacturers by offering detailed and actionable instructions for preparing lidocaine injections from powder form.

Lidocaine powder is the freeze-dried (lyophilized) variant of lidocaine hydrochloride, presented as a sterile, crystalline, and colorless powder. It is manufactured for stability and ease of long-term storage, allowing the lidocaine to remain potent over time without degradation or contamination.
The powder is intended to be dissolved with an appropriate sterile diluent—often sterile water or normal saline—to produce a clear, isotonic injectable solution. This solution facilitates various administration routes including intradermal, subcutaneous, and regional nerve blocks, depending on clinical needs.
Using lidocaine in powder form offers advantages such as extended shelf-life, ability to customize concentrations based on patient or procedural requirements, and compatibility with strict sterility protocols in pharmaceutical production.
The main reasons for reconstituting lidocaine powder are:
- Storage Stability: As a powder, lidocaine remains stable for longer periods at ambient temperatures, reducing degradation.
- Customized Concentrations: Reconstitution enables healthcare providers and manufacturers to prepare solutions with desired lidocaine concentrations tailored to specific anesthesia requirements.
- Ease of Shipping and Handling: Powdered forms take up less space and reduce weight, lowering transportation costs and allowing OCD (Original Content Development) or OEM manufacturers to deliver standardized raw products to global clients.
- Enhanced Control: The ability to mix the anesthetic immediately before use ensures maximum potency and safety by reducing the risks of contamination or chemical change.
Before beginning the reconstitution process, make sure to collect the following:
- A vial containing lidocaine powder (lyophilized lidocaine hydrochloride).
- Sterile diluent: sterile water for injection or preservative-free normal saline. Occasionally, injectable lidocaine at lower concentrations (0.5% or 1%) may serve as a diluent for reducing injection pain.
- Sterile syringe and needles: selection will depend on dilution volume and injection site.
- Alcohol prep pads for disinfecting vial rubber stoppers and injection sites.
- Gloves and aseptic materials to maintain strict sterility.
- Sharps container for proper disposal of used needles and vials.
- Labels or markers to identify reconstituted solution concentration and preparation time.
Start by washing your hands thoroughly and wearing sterile gloves. Ensure the work area is clean, and gather all supplies, verifying sterility and expiration dates. Examine the lidocaine powder vial for any damage or signs of contamination. Prepare a stable surface for the reconstitution workflow that complies with aseptic techniques.
The choice of diluent significantly influences solution compatibility and patient outcomes:
- Sterile Water for Injection: Frequently used as a diluent when creating lidocaine solutions from powder but can cause slight irritation upon injection due to hypotonicity.
- Normal Saline (0.9% Sodium Chloride): Preferred for isotonic solutions, reducing discomfort during injection and maintaining tissue compatibility.
- Diluting with Lower-Strength Lidocaine Solution: Sometimes used to reduce injection pain and achieve desired anesthetic concentration smoothly.
Always consult product-specific or regulatory guidelines before diluent selection.
Calculate the volume of diluent based on the final concentration desired. For instance, if the powder contains 600 mg of lidocaine:
- To prepare a 1% solution (10 mg/mL), add 60 mL of diluent.
- For a 2% solution (20 mg/mL), add 30 mL.
Ensure the total volume and concentration meet your procedural needs. Accurate calculations prevent overdosing or underdosing.
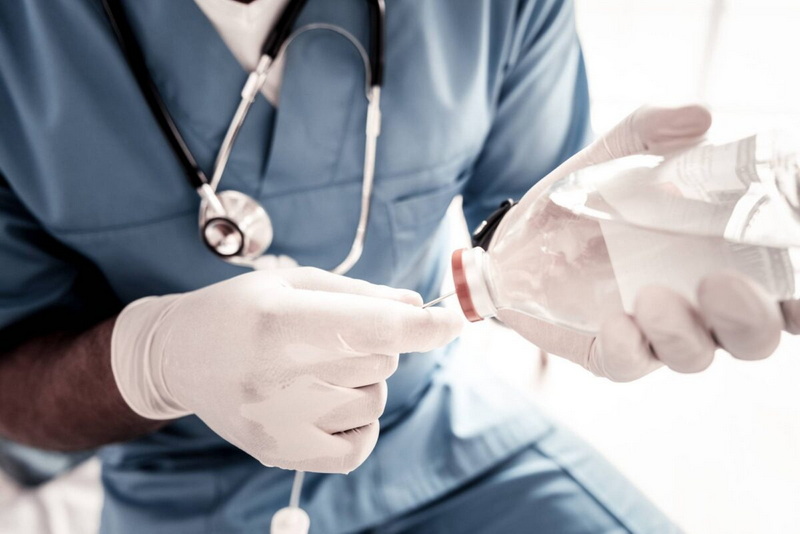
Disinfect the vial rubber stopper thoroughly with alcohol prep pads. Attach a sterile needle and syringe, withdraw the measured amount of diluent, and inject slowly into the vial containing the powder. Inject at an angle to minimize foaming or air bubbles.
Gently swirl or invert the vial to dissolve the lidocaine powder fully. Avoid shaking vigorously to prevent bubble formation, which can affect dosage accuracy. The solution will become clear and free of visible particles once properly dissolved. If undissolved powder remains, do not use the solution.
Use a sterile syringe to draw the reconstituted lidocaine, taking care to expel all air bubbles. Label the syringe with critical information such as drug name, concentration, patient details, and time of preparation.
Administer the injection promptly, adhering to sterile injection protocols. Any remaining solution should be discarded as per institutional or regulatory protocols to avoid contamination. Dispose of needles and vials safely in sharps containers.
- Always reconstitute only the amount needed for immediate use to minimize microbial contamination risks.
- Strictly use aseptic techniques throughout all steps.
- Avoid using lidocaine with epinephrine in areas with compromised blood flow (such as fingers, toes, or ears) due to the risk of ischemic injury.
- Know the maximum safe dosage: Typically, lidocaine's limit without epinephrine is about 4.5 mg/kg body weight, not exceeding approximately 300 mg per adult.
- Confirm patient allergies to lidocaine or similar anesthetics to prevent hypersensitivity reactions.
- Avoid repeated withdrawals from a single vial to preserve sterility.
- If storage of the reconstituted solution is needed, keep it below 25°C and use it within a few hours.
Reconstituted solutions are generally recommended for immediate use but may be stable for several hours under controlled storage conditions. In pharmaceutical manufacturing, quality control tests including sterility assays and potency verification are essential before batch release.
For hospitals or clinics, strict adherence to storage time limits and proper labeling prevents inadvertent use beyond recommended time frames.
For international partners requiring customized lidocaine powders or formulations, OEM services can tailor:
- Concentrations suited to different procedure types.
- Addition of compatible vasoconstrictors like epinephrine in fixed ratios.
- Packaging options for bulk and individual doses.
- Documentation and compliance in accordance with destination country regulations.
Our company offers these OEM services with full technical support, ensuring products meet diverse market and clinical requirements.
Reconstitution of lidocaine powder into an injectable numbing solution is a critical process requiring precision, sterility, and careful handling. Proper technique ensures a safe, effective anesthetic, minimizes patient discomfort, and complies with clinical and regulatory standards. Whether you are a healthcare provider or pharmaceutical manufacturer, understanding these principles safeguards product quality and patient safety.
We specialize in supplying high-quality lidocaine powders and offering professional OEM services tailored to international customers' needs. Contact us to learn how we can support your business with customized lidocaine powder formulations and reliable production capabilities.

Reconstituted lidocaine is ideally used immediately to minimize infection risk. If storage is necessary, keep it under sterile conditions at temperatures below 25°C, and use within a few hours.
Yes, sterile water for injection or normal saline are both acceptable. Saline is generally preferred for patient comfort due to its isotonic nature.
Adding adrenaline (epinephrine) prolongs anesthetic effect and reduces bleeding but should be used cautiously. Avoid using adrenaline-containing solutions in areas with poor blood circulation such as fingers or toes.
Common concentrations are 1% (10 mg/mL) and 2% (20 mg/mL), chosen based on procedure type and duration of anesthesia needed.
For adults, the maximum dose without epinephrine is approximately 4.5 mg/kg body weight (up to 300 mg total). With epinephrine, this limit increases.
[1] https://www.fda.gov/media/175365/download
[2] https://www.youtube.com/watch?v=8ECYuiHFObU
[3] https://www.drugs.com/pro/lidocaine-intradermal-injection.html
[4] http://www.bccdc.ca/resource-gallery/Documents/Educational%20Materials/STI/Med%20Handouts/Ceftriaxone_IM_Administration.pdf
[5] https://www.youtube.com/watch?v=b0iHmHNQd9w
[6] https://medicalguidelines.msf.org/en/viewport/EssDr/english/lidocaine-lignocaine-injectable-16682881.html
[7] https://jpdb.nihs.go.jp/jp14e/14data/Part-I/Lidocaine.pdf
[8] https://www.botoxcosmetichcp.com/content/dam/botox-cosmetic-hcp/documents/BTXC%20How-To%20Reconstitute%20Guide.pdf
[9] https://labeling.pfizer.com/ShowLabeling.aspx?id=4497
[10] https://www.droracle.ai/articles/196876/to-reconstitute-2-g-of-rocephin-what-is-the-quantity-of-lidocaine-to-be-used
[11] https://www.youtube.com/shorts/eau2om6MLfY
[12] https://dailymed.nlm.nih.gov/dailymed/fda/fdaDrugXsl.cfm?setid=ef782e5e-e2d7-465a-99c6-b12611f3158c
[13] https://www.pccarx.com/Blog/2-tips-for-compounding-with-lidocaine-and-lidocaine-hcl
[14] https://www.youtube.com/watch?v=xLEKTIVt2QY
[15] https://www.mayoclinic.org/drugs-supplements/lidocaine-intradermal-route/description/drg-20137337
[16] https://www.cheshireandmerseyside.nhs.uk/media/ri3kevoj/administration-of-ceftriaxone-and-lidocaine-for-gonorrhoea-bpas-v21-signed.pdf
[17] https://wtcs.pressbooks.pub/nursingskills/chapter/5-10-reconstituted-medication/
[18] https://pdf.hres.ca/dpd_pm/00065888.PDF
[19] https://dailymed.nlm.nih.gov/dailymed/fda/fdaDrugXsl.cfm?setid=999dd95d-87fe-12c0-e053-2995a90aa9f7
[20] https://www.youtube.com/watch?v=vuzqIsWFn3o