Content Menu
● Historical and Modern Uses of Procaine
● Understanding the Legal Status of Procaine in the USA
>>> FDA Approval and Prescription Status
>>> Bulk Procaine Powder and Research Use
● Availability and Restrictions in the U.S. Market
● Health and Safety Considerations
● Considerations for Foreign Manufacturers and Suppliers
● Comparisons with Other Local Anesthetics
● Best Practices for Importing Procaine to the U.S.
● Frequently Asked Questions (FAQ)
>> 1. Is it legal to buy procaine powder online in the USA?
>> 2. Can dentists or doctors legally prescribe and use procaine in the U.S.?
>> 3. Is procaine the same as Novocain?
>> 4. Can procaine be legally imported for research purposes in the U.S.?
>> 5. What are the consequences of selling or importing procaine without proper authorization?
The global pharmaceutical, biotechnology, and medical device markets continue to expand rapidly, accompanied by increasing regulatory intricacies. For manufacturers and suppliers, particularly those based in countries like China engaged in the research, production, and OEM supply of specialty chemicals such as procaine, understanding the legal framework of target markets like the United States is essential. Procaine, historically a widely used local anesthetic, raises frequent questions regarding its legal procurement and use in the U.S.
This article thoroughly explores the legal status of procaine within the United States, detailing its history, typical applications, regulatory environment, and best practices for manufacturers, wholesalers, brand owners, and researchers. It also highlights critical compliance issues and the nuanced distinctions between medical, research, and industrial use.
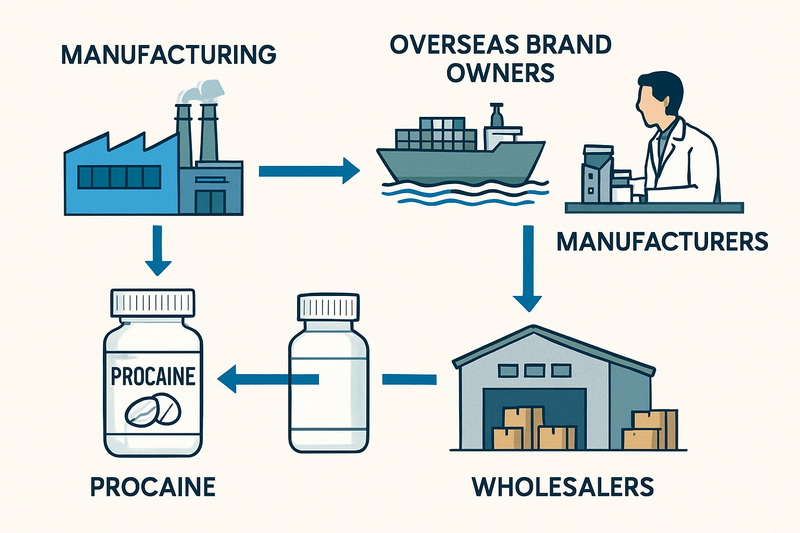
Procaine, commonly recognized as “Novocain,” is a member of the amino ester family of local anesthetics. First synthesized in 1905, it became a breakthrough in pain management, particularly in dentistry and minor surgical procedures. Acting by temporarily blocking nerve impulse transmission, procaine effectively numbs targeted areas, reducing patient discomfort during invasive treatments.
Despite its early prominence, procaine has gradually been replaced in many clinical settings by newer anesthetics with longer duration and improved safety profiles, such as lidocaine and articaine. Nevertheless, procaine remains important in certain applications, including some medical practices, veterinary medicine, and pharmaceutical research settings.
Throughout its clinical history, procaine's principal use has centered on local anesthesia for dental and minor surgical procedures. It was especially popular because of its rapid onset and short duration, which allowed for quick interventions without prolonged numbness.
In addition to human healthcare, procaine finds occasional application in veterinary medicine, where its anesthetic properties assist with animal surgeries and treatments.
Moreover, procaine is utilized in pharmaceutical research and development as a chemical reagent, facilitating studies of nerve function and drug formulation. In this capacity, bulk procaine powder is frequently required by laboratories and manufacturers, which necessitates understanding of regulatory standards for industrial use.
The demand for procaine—while reduced for retail pharmaceutical products in the U.S.—persists in specialized sectors like research, OEM pharmaceutical production, and niche medical applications.
In the United States, the U.S. Food and Drug Administration (FDA) has not approved any procaine hydrochloride products for over-the-counter sale. While procaine was historically available as an injectable anesthetic under brand names such as Novocain, manufacturers have discontinued these products, largely due to market dynamics rather than safety or efficacy concerns.
Procaine is classified as a prescription medication, making it legal only when prescribed or administered by licensed medical professionals within clinical settings. This status means that private individuals are prohibited from buying procaine for personal use without a valid prescription from an authorized healthcare provider.
Unlike many prescription medications, raw procaine powder is not classified as a controlled substance under the federal Controlled Substances Act. This legal positioning makes procaine somewhat unique since possession and sale outside of illicit contexts are generally not criminal offenses at the federal level.
However, bulk procaine is subject to strict regulatory oversight given its potential misuse. Distributors and importers must comply with relevant FDA, Drug Enforcement Administration (DEA), and U.S. Customs regulations. The shipment of pure procaine powder must be accompanied by proper licensing, transparent documentation of the end-use, and thorough compliance with safety and quality standards. Failure to meet these requirements can result in shipment holds, customs seizures, or legal penalties.
While federal laws dictate the overall framework governing controlled substances and drug approval, individual U.S. states may impose additional rules. Notably, some states, such as Nevada, have enacted exceptions permitting pharmacists to dispense procaine hydrochloride without a prescription under specific conditions. These provisions are uncommon and generally require strict record-keeping and compliance verification.
In most states, procaine remains prescription-only, with no authorization for retail or OTC sales. Manufacturers and importers must understand the regulatory nuances of each state where their products will be distributed.
For foreign manufacturers and exporters looking to supply the U.S. market, adherence to import controls is vital. The U.S. Customs and Border Protection (CBP) enforces strict import documentation requirements, mandating:
- Detailed declarations of product contents and purity
- Verification of the consignee's licenses or registrations
- Evidence demonstrating lawful, medical, or research use
Imports lacking these validations risk being detained or rejected. OEM suppliers must also track the supply chain diligently to prevent diversion to unauthorized parties.
The legal availability of procaine in the U.S. varies with its intended application:
- Personal Use: Not legal for purchase or use without a prescription.
- Medical and Dental Use: Legal when prescribed and administered by licensed healthcare professionals.
- Research and Industrial Use: Conditional legality, requiring compliance with federal and state regulations, plus registration/licensing for laboratories or manufacturers.
- Importation for OEM: Permitted with full regulatory adherence, including documentation of the medical or research end-use, product quality verification, and responsible distribution practices.
Though procaine is not a scheduled substance, it remains heavily scrutinized by regulators concerned with pharmaceutical safety and drug diversion prevention.
Use of procaine carries certain risks, especially if handled improperly or used without professional oversight:
- Allergic Reactions: Some individuals experience allergic responses to procaine or its metabolites (notably para-aminobenzoic acid, PABA), which can range from mild skin irritation to severe anaphylaxis.
- Toxicity Risks: Overdosage or inadvertent injection into blood vessels may cause serious side effects, including cardiovascular or respiratory compromise.
- Proper Handling: Bulk powders must be managed in controlled environments, with accompanying Certificates of Analysis (COA) and Material Safety Data Sheets (MSDS) to ensure laboratory and manufacturing safety.
Because of these concerns, only qualified personnel should handle or administer procaine products.
International suppliers intending to export procaine to the U.S. face multiple challenges requiring detailed knowledge of regulations and logistical protocols. Key strategies include:
- Verification of Buyers: Confirm clients hold valid licenses and registrations appropriate for receiving pharmaceutical or research chemicals.
- Complete Documentation: Provide accurate Certificates of Analysis, safety data, and customs paperwork to facilitate smooth importation.
- End-Use Transparency: Clearly state the purpose of the product—medical, dental, research, or OEM manufacturing—to minimize regulatory suspicion.
- Compliance Monitoring: Stay informed of updates in FDA, DEA, and individual state regulations to avoid inadvertent violations.
- Supply Chain Traceability: Maintain comprehensive records linking each shipment to its final user and application, crucial for audits or regulatory inquiries.
These practices protect both supplier reputations and legal standing in the highly regulated U.S. market.
While procaine has historical significance, newer local anesthetics dominate current medical practice due to enhanced efficacy and safety profiles. Compared to agents like lidocaine or articaine:
- Procaine has a shorter duration of action.
- It carries a moderately higher risk of allergic reaction.
- None of these anesthetics are available OTC in the U.S.
Such distinctions influence prescribing patterns and market demand.
For companies supplying procaine to U.S. clients, following a robust procedural framework ensures compliance and reduces business risks:
1. License Verification: Confirm all customers hold valid state and federal professional or institutional licenses before sale.
2. Accurate Labeling and Packaging: Ensure products meet USP quality standards and comply with U.S. labeling laws.
3. Engage Experienced Customs Brokers: Utilize brokers skilled in pharmaceutical imports to navigate complex clearance procedures.
4. Maintain End-User Documentation: Collect and store buyer attestations or contracts confirming product use complies with U.S. laws.
5. Ongoing Regulatory Monitoring: Track legal updates and adapt supply strategies accordingly.
Procaine occupies a specialized but well-defined legal niche within the United States. Although no longer available as a common over-the-counter anesthetic, it remains legally accessible through prescription for medical and dental applications. Bulk procaine powders used in research and OEM manufacturing are also legal, provided all federal, state, and customs regulations are strictly followed.
For foreign manufacturers and suppliers targeting the U.S. market, understanding the multifaceted regulatory environment—spanning FDA approval status, DEA classification, customs inspection requirements, and state-specific rules—is essential. Compliance, transparency regarding end-use, and thorough documentation form the pillars of lawful business operations.
In summary:
- Procaine is legal in the U.S. only under prescription or licensed professional use.
- It is not a controlled substance but subject to rigorous oversight, especially in bulk powder form.
- Unauthorized purchase or importation for personal use is prohibited.
- Suppliers must adhere to all appropriate legal and safety frameworks to ensure successful market access.
With this knowledge, international OEMs, distributors, and researchers can confidently navigate the U.S. regulatory environment relating to procaine.

Generally, no. While procaine powder is not a controlled substance federally, its sale and purchase are regulated and restricted to licensed medical professionals, registered research institutions, or authorized pharmaceutical manufacturers. Purchasing procaine for personal or unlicensed use online is illegal.
Yes. Licensed dentists and physicians can prescribe and administer procaine for medical and dental procedures. Patients cannot directly purchase procaine without a prescription from a licensed practitioner.
Novocain is a brand name historically associated with procaine hydrochloride. Although injectable Novocain has been largely discontinued in the U.S., generic procaine remains recognized and utilized under professional supervision.
Yes. Importation for legitimate medical research or pharmaceutical manufacturing is legal when accompanied by full documentation, compliance with import regulations, and clear demonstration of lawful intended use.
Penalties can include regulatory fines, seizure of shipments, business license suspension, or legal prosecution, especially if the product is diverted for illicit use or sold without medical justification.
Hot tags: Is Procaine Legal In USA, Buy Procaine In The United States, Procaine FDA Status, Purchase Procaine USA, U.S. Anesthetic Legal Guidelines, Legal Procaine Source USA, Can I Buy Procaine Legally USA, U.S. Laws On Procaine, Procaine Availability In USA, U.S. Market For Procaine