Content Menu
● What Is Procaine: Chemical and Pharmacologic Foundations
● Procaine's Mechanism of Action: The Science of Nerve Blockade
● Procaine Absorption: From Injection and Administration to Systemic Circulation
>> Gastrointestinal Absorption: The Role of pH
● The Mechanism: Why Does pH Matter So Much?
● Biotransformation and Elimination
● Special Considerations: Species Differences, Safety, and Drug Interactions
● Pharmaceutical Product Development: Practical OEM/ODM Guidance
● Beyond Basic Anesthesia: Procaine's Broader Applications
● Regulatory and Marketing Considerations
● Innovation in Procaine Delivery Systems
● Frequently Asked Questions (FAQ)
>> 1. What is the main site of procaine absorption within the digestive system?
>> 2. How does pH specifically impact procaine's membrane permeability?
>> 3. What happens to procaine after it is absorbed?
>> 4. Are there risks to procaine administration via oral or non-injection routes?
>> 5. How do pharmaceutical companies improve procaine efficacy in clinical practice?
Procaine remains one of the cornerstone local anesthetics in both human and veterinary medicine, celebrated for its effectiveness, manageable safety profile, and consistency in numbing tissues for numerous clinical, dental, and surgical procedures. For Chinese manufacturers, global OEM/ODM suppliers, and pharmaceutical developers, understanding exactly where procaine is absorbed, how pH dictates its absorption kinetics, and the clinical implications of these properties is essential. This article delivers a comprehensive, science-driven exploration of procaine's absorption journey, the underlying pharmacology, and the opportunities these offer for product development and international business.
Procaine is part of the amino ester class of local anesthetics, renowned as the prototypical compound in its category. Developed in the early 1900s, its structure—a para-aminobenzoic acid derivative with a diethylaminoethyl ester side chain—promotes water solubility and accounts for its comparatively rapid metabolism. Its clinical use is largely attributed to its predictable onset, adequate duration for minor and intermediate procedures, and comparatively low toxicity at regular doses.[1][2][3][9]
Like other local anesthetics, procaine's primary function is to inhibit nerve signal transmission. This is achieved by blocking voltage-gated sodium channels situated along the neuronal cell membrane. The aromatic ring component of the procaine molecule aids in membrane passage, while the ester linkage oversees its rapid hydrolysis in plasma. Once in the neuronal vicinity, procaine diffuses across the neural and epineural membranes, reaching the cytoplasm where it binds to the cytoplasmic segment of voltage-gated sodium channels. This binding preferentially affects active (open) channels, stabilizing them in a nonconductive state and preventing the sodium influx necessary for action potential initiation.[2][3][8]
The nerve fibers affected most rapidly are those transmitting dull pain, temperature, and autonomic information, followed by those driving acute pain and, eventually, motor and proprioceptive signals. A secondary pharmacological effect of procaine is its mild vasodilator action, which can increase its own removal from the site of injection and limit duration—countered clinically by formulation with vasoconstrictors such as adrenaline.[3][6][7]
Most clinical applications deliver procaine via injection, either subcutaneously, intramuscularly, or as part of more complex depot preparations. After parenteral injection, procaine is rapidly absorbed into local capillaries, with peak plasma concentrations achieved in as little as 1–3 hours, depending on the specific formulation, site of administration, and presence of vasoconstrictors. Formulations that contain adrenaline promote vasoconstriction, which slows absorption into systemic circulation, extends local anesthetic effect, and reduces the risk of toxicity.[7][1]
Oral procaine absorption is distinctly limited. As a weak base (pKa ≈ 8.7 at 20°C), procaine is overwhelmingly ionized at the low pH values prevailing in the stomach and proximal small intestine. Since passive diffusion across gastrointestinal membranes requires a non-ionized (uncharged) state, the absorption of procaine is dramatically diminished in acidic environments.[10][11][1]
It is only in the ileum, the last segment of the small intestine—where the environmental pH approaches neutrality or mild alkalinity—that the proportion of non-ionized procaine increases sufficiently to permit meaningful passive absorption. Nonetheless, even in the ileum, the drug's absorption remains modest, and overall oral bioavailability is low. This is compounded by procaine's rapid metabolism in the plasma, meaning most systemically absorbed amounts are quickly hydrolyzed before reaching central circulation.[1][10]
Topical application of procaine to mucous membranes (e.g., oropharyngeal, nasal, or rectal surfaces) leverages the often higher, more neutral local pH to promote non-ionized drug fractions and improved absorption. Here, the mucosa offers a direct route to capillaries that bypass the harsh acidic gastric environment, allowing more efficient systemic uptake for certain clinical uses.[11][12]
The passage of procaine molecules through biological membranes relies on Fick's Law of Diffusion and on the partition coefficient between non-ionized and ionized molecules in different pH environments. The Henderson-Hasselbalch equation quantifies this relationship:
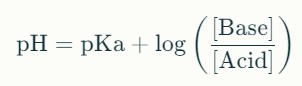
For procaine as a weak base, higher (more alkaline) pH shifts equilibrium towards the non-ionized (base) state, facilitating membrane crossing. In contrast, acidic pH in the stomach or duodenum maintains the molecule in an ionized state, dramatically reducing passive membrane permeability. This explains the much greater absorption rates observed in the ileum, with pH values closer to 7.4 or higher, than in the acidic segments of the gut.[10][11][1]
Once in systemic circulation, procaine is subject to brisk hydrolysis by plasma cholinesterase enzymes. This process converts procaine into para-aminobenzoic acid (PABA) and diethylaminoethanol, both of which are less pharmacologically active than the parent compound. Notably, PABA is relevant for clinical and veterinary medicine since it can antagonize the efficacy of sulfonamide antibiotics. The elimination half-life of procaine in humans is less than one hour, although this may be extended in the presence of enzyme deficiencies, hepatic impairment, or when formulated for sustained release.[9][1]
Diethylaminoethanol, while possessing some weak anesthetic and antispasmodic effects, contributes little to the overall pharmacologic profile in standard clinical concentrations.[1]
Species-specific variations in plasma esterase levels, gastrointestinal pH profiles, and membrane permeability result in significant differences in procaine pharmacokinetics and pharmacodynamics between animals and humans. For pharmaceutical and veterinary OEM/ODM suppliers, grasping these nuances is vital to tailoring products for local regulatory requirements and international safety standards.[1]
Dose-adjustment and careful patient selection are crucial in individuals (or animals) with compromised renal function or genetic defects in plasma cholinesterase, where slower metabolism can lead to increased drug exposure and, rarely, toxicity.
While procaine is generally well tolerated, allergic reactions (often linked to the PABA metabolite) can occur, as can mild CNS or cardiovascular excitability with accidental overdose or rapid intravenous injection. The story underscores the importance of thoughtful formulation, dosing, and patient monitoring.[13][3][9]
For OEM/ODM pharmaceutical and medical device companies, leveraging a deep understanding of procaine's absorption dynamics translates into superior product customizations, competitive market positioning, and regulatory compliance:
- Formulation Choices: Procaine is frequently produced as procaine hydrochloride for water solubility and shelf stability. Adjustments to buffer systems in injectable solutions ensure rapid local anesthetic onset and optimal shelf life but must anticipate rapid neutralization and absorption post-injection.[14][15]
- Combined Preparations: Formulations that integrate vasoconstrictors (adrenaline) can substantially prolong the duration of anesthesia by reducing procaine's local clearance.
- Oral and Specialty Forms: Though oral procaine is rarely indicated due to low bioavailability, innovative product designs such as enteric-coated or sustained-release forms may harness the optimal pH environment of the ileum for specific delivery needs.
- Veterinary Products: Animal health products, especially for equine, bovine, and companion animal markets, often account for unique metabolic and absorption properties through formulation tailoring.
Understanding the influence of formulation pH, salt form, and delivery route helps international suppliers adjust their offerings for regional regulatory and clinical expectations, supporting value-added partnerships with foreign brands and wholesalers.
Although procaine's legacy is local anesthesia, it also finds niche uses in certain neuropharmacologic studies, experimental vasodilator therapies, and in potential CNS-active compound investigations. Its capacity to influence muscarinic cholinergic receptors highlights why understanding site and rate of absorption—especially under conditions where rapid CNS access is necessary—remains relevant even outside classical clinical settings.[5]
In OEM/ODM settings, communicating such versatile application knowledge empowers clients with new product ideas and helps establish authority for Chinese suppliers targeting Western and emerging international markets.
Demonstrating technical mastery of procaine absorption and pH-dependence not only strengthens credibility during regulatory approval processes but also enhances B2B sales and marketing. Educational resources, detailed technical datasheets, and application notes attuned to client needs signal reliability and expertise, key differentiators for Chinese manufacturers entering global supply chains.
Modern drug delivery and advanced pharmaceutical development have inspired novel approaches to optimizing procaine's clinical utility:
- Depot Injections and Sustained Release: Polymeric microspheres and oil-based depots can release procaine (or its combinations, such as procaine penicillin) over extended periods, bypassing rapid plasma hydrolysis and optimizing efficacy in both human and veterinary fields.
- Nanoparticle and Liposome Formulations: Encapsulation shields procaine from harsh gastrointestinal or tissue environments, allowing for targeted release at optimal absorption sites, such as the ileum's more neutral pH region.
- Smart Coatings for GI Targeting: Enteric coatings ensure that orally administered forms bypass acidic stomach segments, releasing the drug for absorption under more favorable intestinal conditions.
For international customers and B2B clients, such innovations demonstrate not only a commitment to quality and efficacy but also an understanding of the global market's evolving clinical requirements.
A deep understanding of where procaine is absorbed and the critical influence of pH on its absorption kinetics is foundational for every professional and manufacturer developing or utilizing procaine-based formulations. From its basic chemistry and nerve-blocking action to its metabolic fate, competitive positioning in global OEM/ODM markets depends on offering scientifically grounded solutions. Partner with a technical leader in procaine manufacturing and unlock the pathways to innovation and commercial success. Reach out to us now to discover cutting-edge custom and bulk solutions for your next pharmaceutical or healthcare project.
Absorption is minimal in the stomach and proximal small intestine, but highest in the ileum, where neutral to mildly alkaline pH settings allow more drug to exist in the non-ionized, membrane-permeable form.[10][1]
Procaine's weak base chemistry means low pH (acidic) settings create near-total ionization, restricting absorption, while higher pH (more alkaline or neutral) increases non-ionized forms and thus passive diffusion across membrane barriers.[11][1]
It is rapidly hydrolyzed by plasma enzymes into para-aminobenzoic acid and diethylaminoethanol, both of which are rapidly excreted. PABA as a metabolite can antagonize sulfonamide antibiotics.[9][1]
Yes—oral bioavailability is poor due to both physicochemical and metabolic barriers, and while mucosal administration is possible, careful dosing and clinical observation are necessary to avoid hypersensitivity or systemic toxicity in susceptible populations.[3][13]
By careful formulation of injections with vasoconstrictors or pH modifiers, and through ongoing research into delivery systems that maximize absorption at optimal GI tract segments or enable prolonged-release patterns for therapeutic benefit.[7][1]
[1](https://www.ema.europa.eu/en/documents/mrl-report/procaine-summary-report-committee-veterinary-medicinal-products_en.pdf)
[2](https://go.drugbank.com/drugs/DB00721)
[3](https://www.ncbi.nlm.nih.gov/books/NBK551556/)
[4](https://synapse.patsnap.com/article/what-is-the-mechanism-of-procaine-hydrochloride)
[5](https://www.nature.com/articles/1300404)
[6](https://emedicine.medscape.com/article/873879-overview)
[7](https://www.sciencedirect.com/science/article/abs/pii/S0099542808606280)
[8](https://pubchem.ncbi.nlm.nih.gov/compound/Procaine)
[9](https://en.wikipedia.org/wiki/Procaine)
[10](https://pubmed.ncbi.nlm.nih.gov/2234105/)
[11](https://www.sciencedirect.com/science/article/pii/0028390872900822)
[12](https://jamanetwork.com/journals/jama/fullarticle/324749)
[13](https://www.webmd.com/vitamins/ai/ingredientmono-391/procaine)
[14](https://jpdb.nihs.go.jp/jp14e/14data/Part-I/Procaine_Hydrochloride_Inje.pdf)
[15](https://dailymed.nlm.nih.gov/dailymed/fda/fdaDrugXsl.cfm?setid=c80c810a-60e0-49bd-139c-95aec3a286fc)
Hot tags: Procaine Absorption Mechanism, pH Effects on Procaine, Procaine Pharmacokinetics, Procaine Ionization pH, Local Anesthetic pH Impact, Procaine Absorption Rate, Procaine Hydrochloride pH, Procaine Efficacy at pH, Procaine and Tissue pH, Procaine Absorption Studies