Content Menu
● Chemical Structure and Amino Groups in Procaine
● Functional Group Behavior and Protonation Dynamics
>> Protonation and Salt Formation
>> pKa Considerations and Basicity
● Synthesis Strategies: Industrial and Laboratory Perspectives
>> Challenges and Quality Control
>> Clinical Use
>> OEM Supply and B2B Opportunities
>> Regulatory and Safety Considerations
● Mechanism of Action and Biological Interactions
>> Ester vs. Amide Local Anesthetics
● Advanced Pharma and Biotech Roles
>> Analytical and Research Uses
● Industry Trends: OEM/ODM Synergy and International Supply
● Expanding Global Pharmaceutical Applications
>> Medical Devices and Combination Products
● Marketing and OEM Advantages for International Clients
● Frequently Asked Questions (FAQ)
>> 1. What two amino functional groups does procaine contain?
>> 2. Which amino group is protonated first in pharmaceutical salt formation?
>> 3. Why does salt formation matter in procaine manufacturing?
>> 4. What are key OEM advantages in sourcing procaine from Chinese suppliers?
>> 5. How does procaine's mechanism of action relate to its chemical structure?
Procaine, best known as Novocaine, is a classic local anesthetic that revolutionized twentieth-century medical and dental care. It is a staple compound in clinics, hospitals, and laboratories worldwide, and the foundation of numerous pharmaceutical products manufactured by advanced Chinese factories serving global brands. Understanding the chemical structure and molecular interactions of procaine is essential for pharmaceutical scientists, quality control professionals, and industrial OEM customers seeking to optimize product performance, purity, and patient outcomes.[2][3]
What makes procaine noteworthy at the molecular level? The answer lies in its two possible amino functional groups, which profoundly influence its physical properties, biological activity, and formulation pathways. This article delivers a comprehensive introduction to these functional groups and their industrial significance, while also discussing synthesis, regulatory considerations, and manufacturing strategies crucial for contemporary exporters and OEM suppliers.
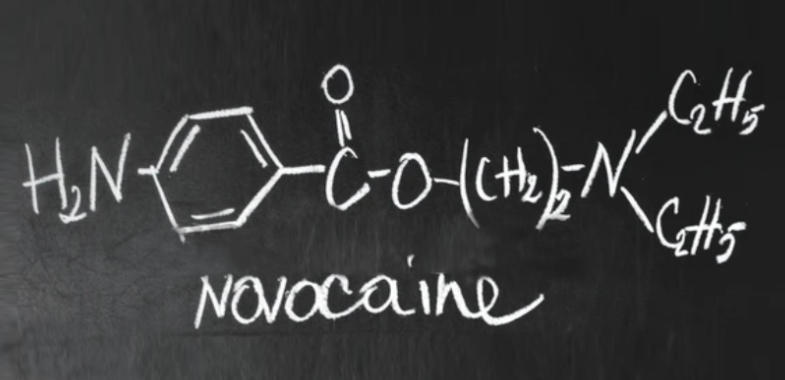
Procaine's molecular formula is C₁₃H₂₀N₂O₂. Its unique configuration contains two amine functional groups that play a direct role in its functionality as a drug.[1][6]
- Primary Amine (--NH₂): This group is attached to the aromatic benzene ring, specifically at the para position (opposite the ester linkage). Its lone pair of electrons is highly available for protonation and reaction.
- Secondary (Diethyl) Amine (--N(C₂H₅)₂): Located within the side chain, this nitrogen atom is bonded to two ethyl substituents, making it bulkier and less reactive compared to the primary amine.[5][6][1]
This essential chemical architecture is responsible for the versatility of procaine in pharmaceutical and medical device applications. The precise placement and nature of these amino groups dictate its solubility, reactivity, and interaction potential with biologically relevant molecules.
In most pharmaceutical products, procaine is administered as a hydrochloride salt. To form this salt, one of procaine's amino groups must accept a proton (H⁺) from an acid, increasing its water solubility and bioavailability for injection or topical use. The protonation process selects the most basic nitrogen atom:[11][5]
- Primary Amine (--NH₂): Highly basic, readily accepts a proton and forms the hydrochloride salt that is used in clinical formulations. This group is generally protonated first because it has a greater tendency to donate electrons compared to the secondary amine.[5]
- Secondary Amine (--N(C₂H₅)₂): While capable of protonation, this group is sterically hindered and less basic due to its alkyl substituents. It is typically not the site of first protonation during salt formation.[5]
The order of protonation is not just a chemical curiosity—it has direct implications for industrial manufacturing, pharmaceutical stability, and regulatory compliance.
The pKa value of the primary amine is usually higher than that of the secondary amine, indicating greater basicity and easier protonation. Understanding these values helps chemists and engineers predict and control salt formation during synthesis, purification, and formulation.[5]
Procaine is synthesized via esterification, joining 4-aminobenzoic acid (which contains the primary amine) to 2-diethylaminoethanol (bearing the secondary amine). Key steps include:[7][2]
- Esterification Reaction: Enables the functional groups to be preserved and precisely positioned.
- Acid-Base Manipulation: Chemists exploit the differential basicity of the amines to control purity and yield.
In OEM factories, production is scaled for mass yields and stringent quality. Process engineers must ensure:
- Consistent Salt Formation: Favoring the primary amine's protonation for optimal product properties.
- Purity and Certification: Maintaining GMP and ISO standards for global distribution, which require rigorous assay of both amino groups.[12][13]
- Purity Verification: Analytical chemistry methods such as HPLC and NMR are used to confirm the molecular composition and correct protonation state.
- Stability Monitoring: Salt stability is assessed under different storage and transport conditions.
Procaine is classified as an aminoester local anesthetic. It is extensively utilized in:
- Dentistry: Numbing the area around a tooth before procedures, with minimal systemic toxicity.[3][14]
- Minor Surgical Procedures: Short-acting local anesthesia for outpatient treatments and wound care.
- Veterinary Medicine: Anesthetic for animals in diagnostic and operative contexts.[6]
Chinese factories such as supplybenzocaine.co.uk provide procaine and derivatives for:
- Global Pharmaceutical Brands: Custom orders for injectable and topical forms.
- Medical Device Manufacturers: Ingredient sourcing for patches, gels, and other delivery systems.
- Bulk Wholesalers: Large-scale procurement for worldwide clinical usage.
- Compliance: Adherence to pharmacopeia and regulatory frameworks (ICH, FDA, EMA, and local standards).
- Toxicology: Controlled risk of hypersensitivity due to the para-aminobenzoic acid segment.
- Pharmacokinetics: Hydrolyzed rapidly in plasma to PABA; excreted via renal elimination with a short half-life.[4]
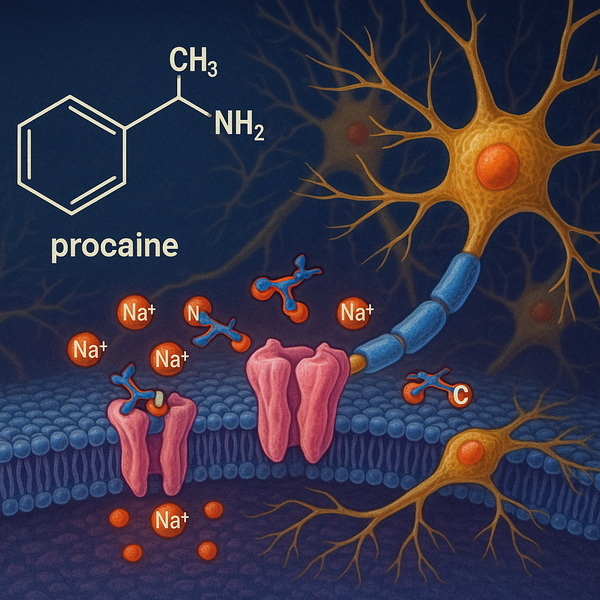
Procaine inhibits sodium influx in neuronal cell membranes by:
- Blocking Voltage-Gated Sodium Channels: Preventing action potential propagation—thus, nerves cannot transmit pain signals.[4][6]
- Receptor Interaction: Also antagonizes NMDA, nicotinic acetylcholine, and serotonin receptor-ion channel complexes, broadening its profile in research pharmacology.
Procaine belongs to the aminoester group. Its rapid hydrolysis and low lipid solubility differentiate it from amide-type anesthetics such as lidocaine, making it the preferred choice for short-acting procedures and allergy-prone patients.[10][6][7]
Scientists may modify either amino group for new derivatives:
- Drug Design: Altering hydrogen bonding and charge to tune pharmacodynamics.
- Specialty Synthesis: Custom molecules for research, diagnostics, and experimental therapies.
- Assay Development: The two amino groups allow for multiple chemical tests and lab protocols.
- Toxicology Research: Procaine is studied as a benchmark for evaluating newer anesthetics.
- High-Yield Manufacturing: Large-scale batch reactors facilitate mass production.
- Customization: OEM factories provide tailored procaine salts suited to diverse end-user requirements, including injection, topical, and compounded formulations.[15][12]
- Certifications: GMP, ISO, and regional regulatory approvals for export.
- Quality Assurance: Documented chemical assays and batch traceability.
- Global Logistics: Rapid shipment and technical support for international buyers.
- Innovation: Collaboration with pharmaceutical developers for next-generation anesthetics.
- Integration: Procaine is compounded with other actives in medical devices, dental gels, and wound care formulations.
- OEM Support: Chinese suppliers offer formulation, packaging, and regulatory consulting across borders.[13][15]
- New Formulations: Inhalable, patch, and sustained-release technologies enhance procaine utility.
- Epigenetic Research: Procaine is studied for potential effects on DNA methylation in cancer therapeutics and regenerative medicine.
Partnering with supplybenzocaine.co.uk and other leading Chinese manufacturers grants access to:
- Scalable Production: From grams to tons, with the ability to match specifications for purity and formulation.
- Competitive Pricing: Reduced manufacturing costs, direct factory partnerships, and flexible order quantities.
- Technical Support: End-to-end assistance in product registration, regulatory compliance, and global logistics.
- Customization Expertise: Unique ability to create modified analogs, custom packaging, and private-label options for pharmaceutical distributors, device makers, and research institutions.
In summary, procaine's two amino functional groups—the primary amine attached to the aromatic ring and the secondary diethylamino side chain—form the molecular foundation for its pharmaceutical use, clinical efficacy, and industrial value. The primary amine, with its greater basicity and preferential protonation, is especially significant for salt formation and injection safety, while the secondary amine contributes to solubility and molecular modification. Whether you are a brand owner, wholesaler, or specialty manufacturer, strategic partnership with certified Chinese OEM suppliers empowers you to secure consistent, high-quality procaine and related compounds at scale.
Contact supplybenzocaine.co.uk today to discover how our custom procaine solutions can elevate your brand, enhance your product portfolio, and meet the demands of global pharmaceutical markets.
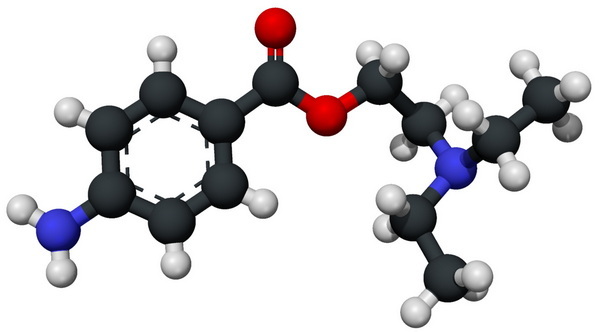
Procaine features a primary amine (--NH₂) attached to the benzene ring and a secondary amine (--N(C₂H₅)₂) within its side chain.[1][5]
The primary amine (--NH₂) is protonated first due to its higher basicity and accessibility, forming procaine hydrochloride for clinical formulations.[5]
Protonation of the primary amine creates a soluble, injectable drug. Salt formation increases bioavailability and product stability.[6][4]
Chinese OEM suppliers offer large-scale production, regulatory certifications, customized formulations, competitive pricing, and technical support for clients worldwide.[12][13][15]
Its amino groups allow procaine to block nerve conduction by interrupting sodium channel activity at cell membranes, underpinning its value as a local anesthetic.[7][4]
[1](https://pubchem.ncbi.nlm.nih.gov/compound/Procaine)
[2](https://www.sciencedirect.com/topics/chemistry/procaine)
[3](https://en.wikipedia.org/wiki/Procaine)
[4](https://go.drugbank.com/drugs/DB00721)
[5](https://www.studocu.com/row/messages/question/6986340/2-which-of-the-two-possible-amino-functional-groups-in-procaine-would-be-protonated-firstwhy3)
[6](https://www.ncbi.nlm.nih.gov/books/NBK551556/)
[7](https://www.sciencedirect.com/topics/pharmacology-toxicology-and-pharmaceutical-science/procaine)
[8](https://homework.study.com/explanation/lidocaine-has-replaced-novocain-procaine-as-the-favored-anesthetic-in-dentistry-which-functional-group-do-the-two-compounds-have-in-common.html)
[9](https://emedicine.medscape.com/article/873879-overview)
[10](https://synapse.patsnap.com/article/what-is-procaine-hydrochloride-used-for)
[11](https://www.studocu.com/en-us/messages/question/5878621/many-drugs-are-administered-as-a-salt-this-increases-the-oral-availability-of-the-drug-procaine-is)
[12](https://www.made-in-china.com/factory/procaine.html)
[13](https://www.made-in-china.com/manufacturers/procaine.html)
[14](https://my.clevelandclinic.org/health/drugs/18945-penicillin-g-benzathine-penicillin-g-procaine-injection)
[15](https://www.made-in-china.com/products-search/hot-china-products/Procaine.html)
Hot tags: Procaine Amino Groups, Procaine Chemical Structure, Procaine Functional Groups, Procaine Uses, Procaine Properties, Procaine Mechanism of Action, Procaine vs Lidocaine, Procaine Anesthetic Effects, Procaine Side Effects, Procaine Applications in Medicine