Content Menu
● Molecular Structure and Molecular Weights
● Why Molecular Weight Conversions Matter
● Molecular Weight Conversion Formula
● Practical Examples in Formulation
● Important Considerations and Tips
● Compatibility Between Lidocaine Forms
● Summary of Key Molecular Properties
● Frequently Asked Questions (FAQ)
>> 1. What is the molecular weight difference between lidocaine and lidocaine HCl?
>> 2. How do I convert from lidocaine base to lidocaine HCl?
>> 3. Can I use lidocaine base directly in injectable formulations?
>> 4. Is the conversion factor always the same?
>> 5. Why is molecular weight conversion important for OEM manufacturers?
Understanding molecular weight conversions between lidocaine base and lidocaine hydrochloride (HCl) is essential for professionals involved in pharmaceutical formulation, research and development, and OEM manufacturing in the biotechnology, medical, and health sectors. Accurate conversions ensure proper dosing, effective drug delivery, and compliance with regulatory standards when supplying overseas brands, wholesalers, and manufacturers.
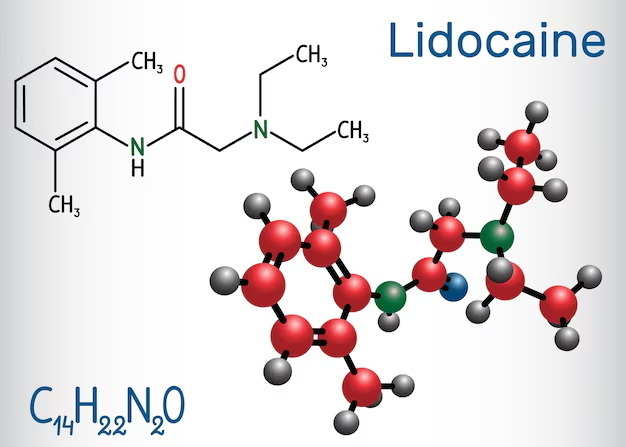
Lidocaine, chemically known as 2-(diethylamino)-N-(2,6-dimethylphenyl)acetamide, is widely used as a local anesthetic and antiarrhythmic agent. Its molecular formula is C14H22N2O, with a molecular weight of approximately 234.34 g/mol. The form commonly used in pharmaceutical products is often lidocaine hydrochloride (lidocaine HCl), which is the hydrochloride salt of lidocaine. This compound has a molecular formula of C14H22N2O·HCl or equivalently C14H23ClN2O, with a molecular weight around 270.8 g/mol.
The molecular weight difference between lidocaine base and lidocaine HCl means that one must perform conversions when switching between the two for compound weighing, drug formulation, or concentration calculations.
Pharmaceutical industries use lidocaine in different chemical forms depending on the formulation needs. Lidocaine base is the pure active compound, but it is often less water-soluble than the hydrochloride salt, which is more soluble and stable for injectable solutions or topical formulations. When purchasing or preparing lidocaine, manufacturers receive it either as the base or the HCl salt, with quantities typically measured by weight.
To ensure consistent drug strength, efficacy, and safety, conversion between lidocaine base and lidocaine HCl is mandatory. This is critical when:
- Calculating doses for various formulations.
- Adjusting order quantities for raw materials.
- Preparing concentrated solutions with precise active ingredient content.
- Labeling products accurately for regulatory compliance.
- Communicating correctly with suppliers and contract manufacturers.
The general formula for converting molecular weights between a base compound and its salt is:

Applying this to lidocaine:
- Molecular Weight of Lidocaine Base (MW_base) ≈ 234.34 g/mol
- Molecular Weight of Lidocaine HCl Salt (MW_salt) ≈ 270.8 g/mol
Example:
To convert 100 mg of lidocaine base to the equivalent amount of lidocaine HCl:
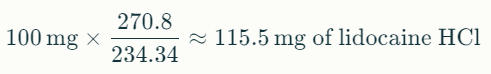
To convert 100 mg of lidocaine HCl to lidocaine base:
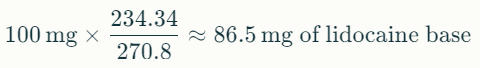
Imagine a manufacturer is producing an injectable anesthetic solution requiring 2% lidocaine base concentration. If using lidocaine HCl raw material, the quantity must be calculated to ensure the final active base concentration is accurate.
- Required lidocaine base: 2 g per 100 mL
- Calculate lidocaine HCl needed:
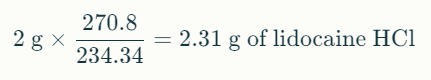
This ensures the formulation delivers 2% active lidocaine base despite using the salt form.
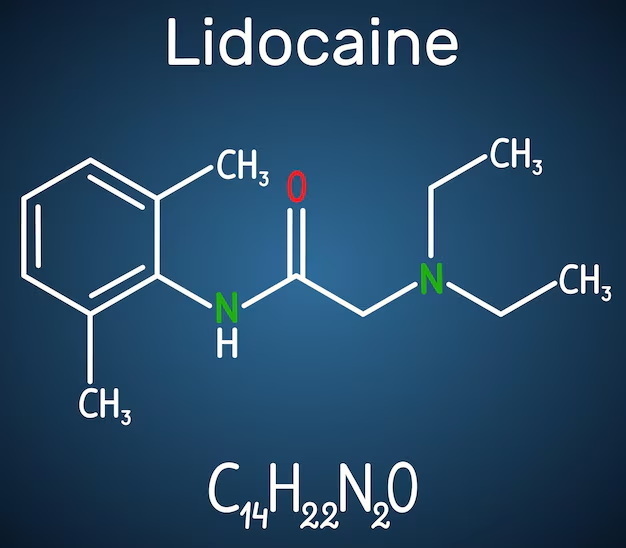
- Always verify the molecular weights from reliable sources such as chemical databases or official pharmacopeias, as minor variations can occur depending on hydration states or purity.
- Confirm the salt form used (free base, HCl, or hydrates), as this affects the molecular weight.
- When working with solutions, consider volume changes due to the salt's presence.
- Use consistent units across calculations, typically milligrams (mg) or grams (g).
- Document calculations carefully for quality control and regulatory audits.
Lidocaine base's lower solubility compared to lidocaine HCl influences formulation development. Pharmaceutical scientists often select the hydrochloride salt for injectable and topical products to improve dissolution and bioavailability. However, when labeling or calculating dosages, conversions between the base and salt forms are essential to reflect the true active drug content.
| Compound | Molecular Formula | Molecular Weight (g/mol) | Solubility |
|---|---|---|---|
| Lidocaine Base | C14H22N2O | 234.34 | Low in water |
| Lidocaine Hydrochloride (HCl) | C14H23ClN2O | 270.8 | High in water |
Performing accurate molecular weight conversions between lidocaine base and lidocaine HCl is indispensable for pharmaceutical manufacturing, ensuring dosage precision and formulation efficacy. As a trusted supplier specializing in biotechnology, pharmaceuticals, and medical devices, your capacity to manage these conversions accurately elevates your service quality for overseas brands and manufacturers.
For consultations or tailored OEM services regarding lidocaine products or other pharmaceutical raw materials, contact us today. Our expert team is ready to assist you with precise formulations, compliance support, and customized solutions to meet your international market needs.
The molecular weight of lidocaine base is approximately 234.34 g/mol, while lidocaine hydrochloride is about 270.8 g/mol due to the added hydrochloride group. This difference necessitates molecular weight conversions for accurate dosing.
Multiply the amount of lidocaine base by the ratio of the molecular weight of lidocaine HCl to lidocaine base:
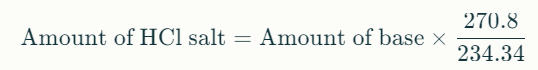
Lidocaine base has poor water solubility, so lidocaine HCl, which is more water-soluble, is commonly used in injectable and topical formulations for better drug delivery.
The conversion factor depends on the exact molecular weights. It may vary slightly if the salt form includes hydrates or other molecules, so always check the specific form of lidocaine used.
OEM manufacturers must ensure the product's active ingredient concentration matches specifications. Incorrect conversions can lead to ineffective or unsafe products and regulatory non-compliance.
Hot tags: Molecular Weight Lidocaine, Lidocaine HCL Conversion, Lidocaine Molecular Weight, Lidocaine HCL Molecular Weight, Lidocaine vs Lidocaine HCL, Lidocaine HCL Dosage, Lidocaine HCL Properties, Lidocaine HCL Uses, Lidocaine HCL vs Lidocaine, Lidocaine HCL Calculation