Content Menu
● Composition and physical properties
● Pharmacology and mechanism of action
● Clinical applications and dosing considerations
● Special populations and considerations
● Manufacturing standards, quality control, and regulatory compliance
● International regulatory considerations for OEM partnerships
● Advanced delivery systems and packaging
● Quality assurance and testing protocols
>> A robust QA program for lidocaine HCL injection includes:
>> Global market opportunities and OEM partnerships
>> Manufacturing technologies and process optimization
>> Safety considerations and risk management
>> Global supply chain and export readiness
>> Clinical and practical considerations for OEM partners
● Important caution for readers and practitioners
● FAQ
>> 1) What is the typical onset time for Lidocaine HCL Injection USP 10 mg/mL?
>> 2) What is the maximum safe dosage for Lidocaine HCL Injection in adults?
>> 3) How long does the anesthetic effect last for 1% lidocaine injection?
>> 4) What are key contraindications for Lidocaine HCL Injection?
>> 5) What standards must OEM manufacturers meet for lidocaine HCL injection production?
Lidocaine Hydrochloride Injection USP 10 mg/mL is a widely used local anesthetic designed for rapid onset of anesthesia during local infiltration, regional nerve blocks, and certain central neural block procedures. As a contract manufacturer serving international brands and distributors, you will appreciate how this formulation balances efficacy, safety, and versatility across multiple clinical settings. This article provides a thorough overview of composition, mechanism of action, clinical applications, manufacturing standards, safety considerations, market opportunities, and practical OEM considerations for global supply.
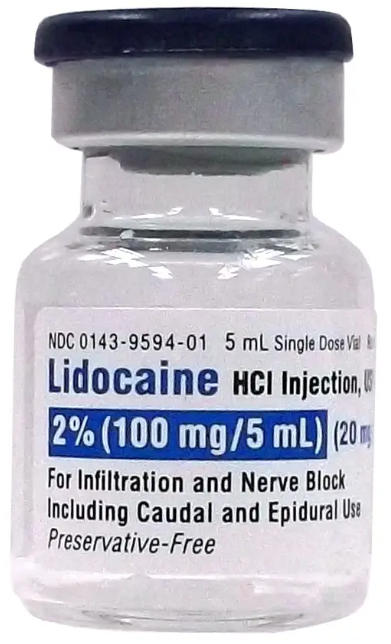
Lidocaine hydrochloride is the active pharmaceutical ingredient (API) in this injection, typically formulated as a sterile solution containing lidocaine hydrochloride at a concentration of 10 mg/mL (1%). The amide-type local anesthetic is widely recognized for its predictable onset and duration in diverse procedures. The product is manufactured to meet USP (United States Pharmacopeia) standards, ensuring potency, purity, and quality control across batches. The API's chemical profile and pharmacokinetic behavior are well characterized, enabling healthcare providers to plan anesthesia strategies with confidence. For OEM manufacturers, maintaining strict lot traceability, aseptic conditions, and validated filling processes is critical to meet international regulatory expectations.
Lidocaine works primarily by stabilizing the neuronal membranes and inhibiting sodium channels, which prevents the initiation and propagation of nerve impulses. This action yields reversible loss of sensation in the targeted region without causing systemic anesthesia. The onset of anesthesia is typically rapid, often within minutes, depending on the administration route and tissue vascularity. Duration of effect varies with dose, technique, and patient factors, commonly ranging from about 1 to 3 hours for standard infiltration and nerve block procedures. The predictable pharmacodynamic profile supports a wide range of clinical applications and makes lidocaine a staple in anesthetic practice. For manufacturers, thorough understanding of stability under various storage conditions and compatibility with delivery systems (vials, prefilled syringes, cartridges) is important to ensure consistent performance.
Local infiltration and peripheral nerve blocks are the mainstay indications for lidocaine HCL injections:
- Local infiltration: Used to numb a defined area for minor procedures such as dental work, skin lesion excision, or suture placement. Typical dosing ranges facilitate adequate anesthesia while maintaining safety margins.
- Peripheral nerve blocks: Employed for targeted anesthesia of larger regions or specific limbs; examples include brachial plexus blocks, intercostal blocks, and regional injections around major nerves. Dosing is tailored to procedure type, site, and patient weight.
Dosing guidelines (illustrative ranges for a typical adult patient) emphasize careful calculation of dose per body weight and maximum allowable total dose to minimize systemic toxicity risks. For OEM and contract manufacturing contexts, aligning packaging and labeling with these range expectations helps ensure safe and effective use across diverse markets. Always consult current prescribing information and regulatory guidelines for precise, region-specific dosing before clinical use.
Dosing and monitoring require special attention in populations with altered pharmacokinetics or physiology:
- Elderly patients: May require dose reductions due to decreased clearance and altered distribution.
- Pediatric patients: Weight-based dosing with strict maximums to reduce toxicity risk.
- Hepatic impairment: Reduced metabolism necessitates dose adjustments and closer observation.
- Cardiac disease: Cautious use and monitoring due to potential cardiovascular effects.
- Pregnancy and lactation: Perioperative analgesia considerations guided by risk-benefit assessment.
Manufacturers supplying lidocaine HCL injection positions themselves within a high-stakes regulatory landscape. Essential elements include:
- USP compliance: Ensuring potency, purity, sterility, and proper physicochemical characteristics.
- GMP (Good Manufacturing Practice): Documented processes, validated equipment, proper facility controls, and comprehensive quality assurance.
- Sterility and endotoxin testing: Routine microbiological testing and validation of sterile processes.
- Container-closure integrity: Guarantee of sterility and containment throughout the product's shelf life.
- Stability and storage: Data supporting shelf life under specified temperatures and light exposure conditions.
- FDA, EMA, and Health Canada pathways: Depending on target markets, regulatory submissions, quality systems, and product registrations will vary.
- ICH guidelines: Harmonized standards for development and manufacturing processes support global market access.
- Country-specific certifications: ISO 13485 for medical devices in some contexts, along with national pharmacopoeial requirements.

Innovations in packaging and delivery systems offer advantages in integrity, dosing accuracy, and convenience:
- Pre-filled syringes and single-dose formats: Improve dosing accuracy and reduce contamination risk.
- Multi-dose vials with sterile closures: Suitable for high-volume settings, with appropriate preservative considerations.
- Cartridges and automated devices: Enable compatibility with electronic or robotic administration systems.
- Packaging materials: Selected to maximize stability and minimize interactions with the formulation.
- Analytical testing: High-performance liquid chromatography (HPLC) for potency and impurity profiling.
- Microbiology: Sterility testing and endotoxin assessment to exclude microbial contamination.
- Physical tests: Clarity, color, and particulate matter evaluation.
- Stability studies: Real-time and accelerated testing to establish shelf life.
- Packaging validation: Container-closure integrity and compatibility testing.
- Growing demand: Driven by expanding surgical procedures, pain management needs, and the shift toward outpatient care.
- OEM advantages: Chinese manufacturers offer scale, cost efficiency, and regulatory know-how to support international brands.
- Regulatory support: Assistance in navigating multiple markets' registration processes and quality compliance.
- Process validation: Documented, reproducible manufacturing steps with critical parameter controls.
- Continuous improvement: Ongoing evaluation of raw materials, energy efficiency, and waste minimization.
- Quality-by-design (QbD): A systematic approach to product and process design to ensure consistent quality.
- Traceability: Comprehensive batch records and material provenance to support audits and recalls if necessary.
- Toxicity prevention: Adherence to maximum recommended doses per body weight and total dose limits.
- Monitoring guidelines: Vigilant observation for signs of local and systemic toxicity during and after administration.
- Contraindications: Known hypersensitivity to lidocaine or related amide anesthetics, severe conduction disorders, and infection at the injection site.
- Special population precautions: Adjustments for age, hepatic function, cardiac conditions, and pregnancy considerations.
- Raw material sourcing: Verified suppliers for lidocaine HCl with consistent quality.
- Documentation: Accurate labeling, certificates of analysis (COA), and regulatory documents for each target market.
- Logistics: Temperature-controlled storage and shipping to preserve product integrity.
- Compliance: Adherence to import/export controls, drug master files, and country-specific advertising and labeling rules.
- Customization options: Ability to tailor concentrations, packaging formats, and labeling to meet partner requirements.
- Regulatory liaison: Support for regulatory submissions and post-market surveillance in different jurisdictions.
- Technical support: Access to formulation development, stability data, and quality assurance expertise.
- Supply reliability: Scalable production capacity, pragmatic lead times, and risk-mitigation strategies.
Lidocaine HCL injection is a potent local anesthetic with systemic toxicity risk if misused. Accurate dosing, sterile technique, and appropriate monitoring are essential for patient safety. Healthcare professionals and OEM partners should rely on current, official prescribing information for dose limits, indications, contraindications, and administration instructions.
Lidocaine HCL Injection USP 10 mg/mL remains a foundational option in local and regional anesthesia due to its rapid onset, reliable duration, and broad applicability across procedures. For international brands and distributors seeking a dependable OEM partner, Chinese manufacturers with strong compliance records, robust QA programs, and deep expertise in sterile injectable production offer competitive advantages, including scale, cost efficiency, and regulatory navigation capabilities. By aligning with a capable OEM partner, brands can access high-quality lidocaine HCL injection that meets stringent global requirements while enabling flexible delivery formats to accommodate diverse clinical settings.
If you are seeking a trusted OEM supplier for Lidocaine HCL Injection USP 10 mg/mL, reach out to discuss your specification, regulatory pathway, and supply timeline. Our team can tailor solutions to your market needs, provide comprehensive documentation packages, and ensure reliable worldwide distribution for your customers. Contact us to know more!
- Onset is usually within 1-5 minutes depending on the administration route and site of injection, with infiltration procedures often showing effects sooner than nerve blocks. [reference: standard dosing guidelines and prescribing information]
- For healthy adults, the single-dose limit is generally 4.5 mg/kg body weight, with a total dose often not exceeding 300 mg for many procedures; nerve blocks may have higher regional caps depending on site and technique. Always consult current labeling.
- Local infiltration commonly lasts about 1.5 to 3 hours; nerve blocks can extend to 1-2 hours or longer depending on dose and tissue factors; digital blocks typically provide 1.5-2 hours of anesthesia.
- Hypersensitivity to lidocaine or other amide local anesthetics, severe conduction disorders, and infection at the injection site; caution in hepatic impairment, elderly patients, and those with cardiovascular disease.
- Compliance with USP potency and purity requirements, sterility and endotoxin controls, proper pH and stability, GMP practices, and applicable international registrations (FDA/EMA/Health Canada) plus relevant ISO certifications.
[1](https://dailymed.nlm.nih.gov/dailymed/fda/fdaDrugXsl.cfm?setid=3b2dd84e-cee8-4e58-d494-395a65a353a0)
[2](https://webfiles.pfizer.com/file/01JAW7D4NFJA5T4974MFX0SHFA)
[3](https://dominapharm.com/product_data.php?id=222)
[4](https://www.pfizermedical.com/lidocaine/dosage-admin)
[5](https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/006488Orig1s100lbl.pdf)
[6](https://dhpp.hpfb-dgpsa.ca/dhpp/resource/90671)
[7](https://www.pfizermedical.com/lidocaine-2/clinical-pharmacology)
[8](https://www.webmd.com/drugs/2/drug-2358-8170/emla-topical/lidocaine-prilocaine-cream-topical/details)
Hot tags: Lidocaine Injection, Lidocaine HCL, Local Anesthesia, Pain Management, Anesthetic Injection, Lidocaine Dosage, Medical Uses of Lidocaine, Side Effects of Lidocaine, Lidocaine Hydrochloride, Injection Safety Guidelines