Content Menu
● Procaine: Chemical Nature and Mechanism of Action
● Formulation Forms and Handling
>> Media prompts for this section:
● Historical Context and Current Landscape
● Regulatory and Compliance Considerations for OEM Partners
● Quality Assurance for OEM Procaine Products
>> Visuals to accompany this section:
● OEM Collaboration Models and Customization Options
● Safety, Ethics, and Patient Considerations
● Practical OEM and Distribution Guidance
>> Media plan:
● Conclusion and Call to Action
● FAQ
>> 1) What is procaine, and in what contexts is it used for injections?
>> 2) What considerations are essential when selecting procaine for OEM production?
>> 3) Which QA tests should be included in a procaine injectable product release?
>> 4) How should regulatory documentation be structured for overseas OEM campaigns?
>> 5) What strategies improve safety and compliance in international distribution?
Procaine is a local anesthetic historically used to block nerve conduction to enable painless procedures. In modern practice, its role has evolved alongside advances in anesthetic pharmacology, control of vasodilation, and the growing emphasis on high-quality, regulated supply chains for medical products. For international brands, contract manufacturers, and distributors, understanding procaine's properties, regulatory considerations, and QA requirements is essential for evaluating OEM opportunities, developing compliant formulations, and ensuring reliable access to ingredients and finished products across markets. This article provides a comprehensive, media-enhanced reference that aligns technical detail with practical OEM considerations, including detailed guidance on quality assurance, regulatory compliance, packaging, and supplier qualification. Throughout, readers will encounter media-ready prompts and placeholders to help integrate illustrative assets such as mechanism diagrams, process flows, and lab visuals into marketing and technical documentation.
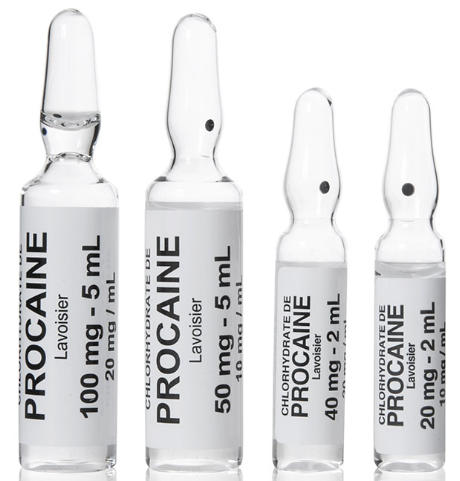
Procaine, an ester-type local anesthetic, acts primarily by inhibiting voltage-gated sodium channels on neuronal membranes, thereby preventing action potential propagation along nerve fibers. This mechanism yields reversible numbness in targeted areas, enabling safe surgical and diagnostic procedures. In OEM contexts, the importance lies not only in pharmacology but also in formulation considerations: stability of the ester bond, compatibility with co-administered vasoconstrictors such as epinephrine where indicated, and the formulation's impact on onset time and duration of anesthesia. Visual assets to support this section may include:
- A schematic diagram showing the site of action on the nerve membrane.
- A chemical structure illustration highlighting the ester linkage and p-aminobenzoate moiety.
- A flow diagram linking mechanism to clinical outcomes (onset, duration, and safety profile).
Procaine is typically supplied for medical use in sterile, injectable forms suitable for precise dosing. For OEM and distribution purposes, attention to sterility, particulate matter control, and packaging integrity is essential. Common considerations include:
- Sterility assurance: aseptic presentation in vials or pre-filled syringes with validated sterilization methods.
- Onset and duration variability: influenced by concentration, presence of vasoconstrictors, and patient-specific factors; formulations may be tailored to regional practice standards within regulatory allowances.
- Stability and storage: defined temperature ranges, protection from light if applicable, and shelf-life determined by rigorous stability testing under accelerated and real-time conditions.
- Close-up images of sterile vials, ampoules, and packaging configurations.
- Short explainer video illustrating correct handling steps from receipt through administration logistics.
Procaine has a long history as a dental anesthetic and for regional/local anesthesia, with its usage evolving as newer agents with improved safety and efficacy profiles emerged. In contemporary practice, procaine remains a reference standard in some regions but is increasingly complemented or replaced by agents such as lidocaine due to favorable pharmacokinetics and allergenicity profiles. For OEM stakeholders, this historical context informs market expectations, regulatory acceptability, and potential repurposing or niche applications within clinical protocols. Media ideas for this section include:
- A timeline graphic highlighting key milestones in procaine's clinical use.
- An archival photograph or schematic depicting traditional dental anesthesia approaches (cleared for reuse and compliant with rights).
Working with procaine-containing products in an OEM setting requires careful navigation of global regulatory frameworks, quality systems, and post-market obligations. Key themes include:
- GMP/ISO certification alignment: ensuring suppliers and manufacturers meet recognized standards for pharmaceuticals and medical devices, with traceability from raw materials to finished products.
- Documentation and batch traceability: robust lot numbering, certificates of analysis, and complete material safety data in each shipment to overseas partners.
- Pharmacovigilance and safety reporting: systems for adverse event reporting, periodic safety updates, and post-market surveillance plans appropriate to target markets.
- Impurity profiling and residuals: rigorous testing to quantify impurities, residual solvents, and potential contaminants, with clear acceptance criteria.
- Packaging and labeling: compliance with international regulations, including multilingual labeling where required and accurate indications of strength, storage, and handling.
- A flowchart detailing the supplier qualification and batch release process.
- A short QA lead interview video (captioned) about supplier audits and documentation controls.
QA is a cornerstone of successful OEM partnerships in biotech and pharma. Essential QA domains include:
- Identity, purity, and potency: establishing validated analytical methods to confirm the chemical identity and pharmacological potency of procaine in each lot.
- Sterility and pyrogen testing: ensuring products are free from microbial contamination and pyrogens.
- Endotoxin assessment: critical for injectable products, especially in multi-use or cross-market distribution scenarios.
- Packaging integrity and leakage prevention: ensuring primary and secondary packaging meet integrity specifications to maintain sterility and potency during transport.
- Stability testing: real-time and accelerated studies to determine shelf life under varied environmental conditions encountered in international supply chains.
- Lab bench shots of analytic instrumentation (e.g., HPLC or GC setups) with non-sensitive data placeholders.
- A labeled schematic of a packaging integrity test (drop test, leakage test, etc.).
- A brief process video showing chain-of-custody controls in a distribution center.
OEM partnerships offer a spectrum of customization possibilities to fit diverse market needs:
- Concentration and dosage formats: adjustable strengths, single-dose presentations, or multi-dose formats tailored to regional preferences and regulatory allowances.
- Co-formulations and combinations: compatibility with vasoconstrictors or other adjuvants where clinically appropriate and compliant with local regulations.
- Packaging configurations: vials, pre-filled syringes, or unit-dose packaging with tamper-evident seals, including barcoding for traceability.
- Labeling and regulatory support: multilingual label development, regulatory submissions support, and product registration pathways for target markets.
Media prompts:
- A comparison chart of packaging options and labeling requirements.
- An overview video illustrating the end-to-end OEM collaboration journey from specification to finished product.
Any discussion of anesthetics must foreground safety, ethics, and patient protection. Topics to cover include:
- Safe handling and administration: training requirements, needle safety, disposal protocols, and facility standards for aseptic technique.
- Informed consent and clinical governance: ensuring patient awareness of procedures and adherence to clinical governance frameworks.
- Ethical supply chain practices: responsible sourcing, conflict-free supply chains, and preventative measures against counterfeit or adulterated products.
Media assets:
- Safety posters and compliance checklists suitable for clinical and manufacturing environments.
- Short training clips demonstrating proper handling and disposal practices.
For international OEM programs, practical guidance helps translate technical knowledge into actionable pathways:
- Supplier qualification: define criteria for supplier audits, certifications, and ongoing oversight.
- Change control: robust processes to handle formulation changes, packaging updates, and labeling modifications across markets.
- Import/export and regulatory navigation: proactive planning for customs, licensing, and market-specific regulatory approvals.
- Quality agreements: specify responsibilities, remediation plans, and dispute resolution mechanisms between partners.
- A flow diagram of the end-to-end supply chain from raw material to finished product, with role annotations.
- A short explanatory video on the OEM engagement lifecycle.
Procaine-containing OEM opportunities exist where providers can deliver reliable supply, rigorous QA, and regulatory-compliant packaging and labeling across international markets. By aligning with a partner that emphasizes transparency, traceability, and proactive regulatory support, foreign brands and distributors can reduce time-to-market and strengthen patient safety. For manufacturers seeking to explore a collaboration, a direct consultation can clarify product specifications, regulatory pathways, and required QA documentation. Contact us to begin a tailored, risk-managed stepwise program for your market entry or expansion.
To support quick reference, the following FAQ addresses common questions from prospective OEM partners and distributors. Each item includes concise, practice-oriented guidance suitable for technical decision-makers in biotech, pharmaceutical, and medical device sectors.
Procaine is an ester local anesthetic used to induce local anesthesia during procedures such as dental work and minor surgical interventions, with potential applications in regional nerve blocks where appropriate and compliant.
Critical factors include supplier qualification, validated potency and purity assays, sterility assurance, stability under target storage conditions, and alignment with regulatory labeling and safety requirements in the intended markets.
Required tests typically encompass identity and purity assays, sterility testing, endotoxin assessment, pyrogen checks, particulate matter evaluation, and stability testing under defined conditions.
Ensure complete batch records, Certificates of Analysis, material safety data sheets, and regulatory-facing labeling, with a clear mapping to each target market's registration and pharmacovigilance requirements.
Implement end-to-end quality management, rigorous supplier audits, standardized change control, comprehensive training, and proactive post-market surveillance to address safety signals promptly.
[1](https://www.webmd.com/vitamins/ai/ingredientmono-391/procaine)
[2](https://en.wikipedia.org/wiki/Procaine)
[3](https://www.rxlist.com/supplements/procaine.htm)
[4](https://go.drugbank.com/drugs/DB00721)
[5](https://medlineplus.gov/druginfo/meds/a615052.html)
[6](https://my.clevelandclinic.org/health/drugs/18945-penicillin-g-benzathine-penicillin-g-procaine-injection)
[7](https://www.ncbi.nlm.nih.gov/books/NBK551556/)
[8](https://go.drugbank.com/drugs/DB09320)
[9](https://www.mims.com/hongkong/drug/info/procaine?mtype=generic)
Hot tags: Procaine Injection, Local Anesthetic, Pain Relief, Procaine Uses, Procaine Side Effects, Procaine Therapy, Procaine Dosage, Procaine Benefits, Procaine Penicillin, Procaine Mechanism