Content Menu
● What is Procaine Penicillin Injection?
● How It Works: Mechanism and Pharmacology
● Clinical Indications and Applications
● Manufacturing, Quality, and Regulatory Considerations for OEM Partners
● Packaging, Labeling, and Distribution Considerations
● Safety, Contraindications, and Adverse Effects
● Clinical Administration and Dosing Guidance (General Context)
● OEM Partnership Value Proposition
● Visuals and Multimedia Integration (Guidelines for Your Website)
● FAQ
>> 1. What infections can Procaine Penicillin Injection treat?
>> 2. How is the injection administered?
>> 3. What are common side effects and allergy considerations?
>> 4. Why is procaine combined with penicillin?
>> 5. How can a company order OEM Procaine Penicillin from your factory?
Procaine Penicillin Injection represents a long-standing, reliable beta-lactam antibiotic formulation used to treat a variety of bacterial infections. When sourced through a capable OEM manufacturing partner, it can be packaged and labeled to meet international market requirements while maintaining high standards of quality, traceability, and regulatory compliance. This article explains what Procaine Penicillin Injection is, how it works, typical clinical applications, and why a modern Chinese OEM facility can serve foreign brands, wholesalers, and manufacturers with customized solutions. It also outlines packaging, quality assurance, regulatory considerations, and practical guidance for collaboration.
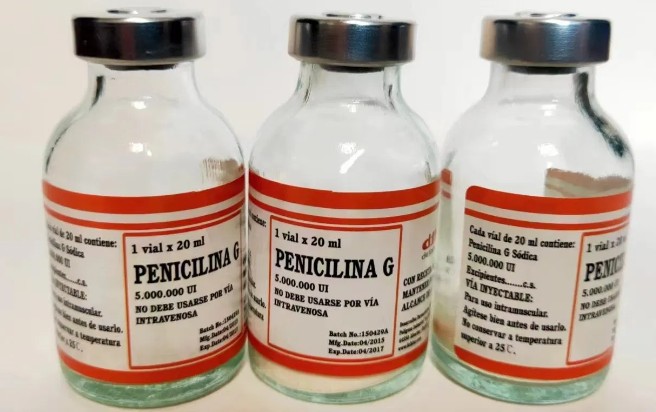
Procaine Penicillin Injection combines penicillin G with the local anesthetic procaine to form a suspension intended for intramuscular administration. The procaine component reduces injection discomfort and slows the release of penicillin G, providing a depot effect that yields prolonged antibacterial action. This combination enables sustained therapeutic levels over a defined dosing interval, making it suitable for certain moderate-to-severe infections where a reliable, extended-interval antibiotic is desirable. Clinically, penicillin G procaine is one member of the broader penicillin family used against susceptible organisms, with established uses in historical and ongoing infectious disease management. For OEM contexts, the drug product is produced under strict GMP conditions, with validated manufacturing processes, stability data, and documentation suitable for international distribution and brand customization.
Penicillin G, the active component, disrupts bacterial cell wall synthesis by inhibiting penicillin-binding proteins. This impairs peptidoglycan cross-linking, leading to bacterial lysis and death in susceptible organisms. The procaine salt delivers a local anesthetic effect to ease the injection experience and—crucially for manufacturing and clinical practice—a depot-release profile that prolongs blood levels of penicillin G. The pharmacodynamic profile supports once-daily to twice-daily dosing in certain regimens, depending on infection type and patient factors. In OEM production, achieving consistent depot characteristics requires precise formulation, sterility, and batch-to-batch validation to ensure uniform release and therapeutic effectiveness across lots.
Penicillin G procaine injections have historic and ongoing relevance for a range of bacterial infections and prophylactic uses. Typical indications include:
- Moderate-to-severe skin and soft tissue infections
- Upper and lower respiratory tract infections
- Erysipelas and scarlet fever presentations
- Certain streptococcal infections
- Prophylaxis and management considerations in rheumatic fever
- Specific cases of syphilis and related infections in regions where penicillin remains a first-line option
In modern practice, the choice of Procaine Penicillin must align with local susceptibility patterns, regulatory approvals, and patient-specific factors. For OEM collaborations, it's essential to maintain robust pharmacovigilance, clear contraindications (notably penicillin allergy), and access to up-to-date clinical guidelines to support pharmacy and medical teams using the product.
A high-quality OEM producer can support foreign brands and wholesalers with comprehensive capabilities:
- GMP-compliant manufacturing: Demonstrates adherence to good manufacturing practices, with validated processes, clean facilities, and controlled environments to ensure product integrity.
- Purity and potency: Rigorous raw material testing, in-process controls, and final product testing to verify potency (units per vial), sterility, particulates, and stability.
- Packaging customization: A range of primary packaging formats (vials or ampoules, with sterile suspensions) and secondary packaging (cartons, inserts, labels) tailored to target markets.
- Documentation: Comprehensive certificates (GMP, ISO), batch records, COAs (certificate of analysis), stability data, and regulatory dossiers enabling rapid market entry.
- Serialization and traceability: Traceable lot numbering, expiry dating, and anti-counterfeiting features as required by different jurisdictions.
- Regulatory support: Regulatory intelligence, labeling alignment with local regulations, and guidance on import requirements, pharmacovigilance frameworks, and post-market obligations.
- Packaging formats: Sterile suspension in robust, tamper-evident containers designed for reliability during transport; consideration for cold-chain requirements if applicable.
- Labeling: Clear strength information, lot numbers, expiry dates, storage conditions, and regulatory approvals referenced as required by the destination market.
- Documentation: Included product inserts, safety data, and compliance documentation to facilitate customs clearance and pharmacovigilance.
- Common adverse effects may include injection-site pain, local swelling, and mild systemic reactions.
- Severe allergic reactions (anaphylaxis) are possible with penicillin-class antibiotics and require immediate medical attention.
- Contraindications include known hypersensitivity to penicillin G, procaine, or related compounds and a history of anaphylactic reactions to beta-lactam antibiotics.
- Caution is warranted in patients with renal impairment or concomitant nephrotoxic therapies; clinical judgment and monitoring are essential.
- Route: Intramuscular injection, administered by qualified healthcare professionals in appropriate clinical settings.
- Typical dosing: Dosing regimens are infection-specific and should be guided by clinical guidelines and susceptibility data; in manufacturing terms, provide clear, market-appropriate dosing ranges and accompanying clinical dosing recommendations for distributors.
- Administration considerations: Ensure aseptic technique, proper needle selection, and injection site management to minimize complications.
- Global reach with localized support: A Chinese OEM facility can offer regulatory documentation, labeling in multiple languages, and region-specific packaging to ease market access.
- Flexible volumes and lead times: Scalable manufacturing with consistent supply for new product launches or steady-state demand.
- Quality assurance as a differentiator: Transparent QA processes, batch testing, stability studies, and traceable supply chains build brand trust.
- End-to-end services: From formulation adjustments (where applicable) to packaging design, labeling, and distribution logistics, the OEM partner can handle multiple facets of product readiness.
- Visuals greatly enhance comprehension and trust. Use high-resolution product images showing the sterile suspension, vials, labeling examples, and packaging configurations.
- Video content can include:
- A short demonstration of the packaging line and quality control checks
- A clinical-use overview illustrating proper administration techniques (performed by a trained professional)
- A manufacturing facility tour highlighting GMP-compliant processes
- For each visual or video, include descriptive alt text, captions that emphasize QA/Regulatory compliance, and any region-specific labeling nuances.
Procaine Penicillin Injection remains a clinically relevant antibiotic with a longstanding track record of effectiveness when used under appropriate medical supervision and regulatory oversight. For foreign brands, distributors, and manufacturers seeking reliable OEM partnerships, a modern, GMP-compliant facility can deliver consistent product quality, customized packaging, rigorous documentation, and robust regulatory support to meet global market requirements.
If your organization is looking to source Procaine Penicillin Injection with reliable supply, flexible OEM packaging, and comprehensive regulatory documentation, contact the OEM team today. Request specifications, sample packaging options, and a tailored quote that aligns with your market strategy and compliance needs.
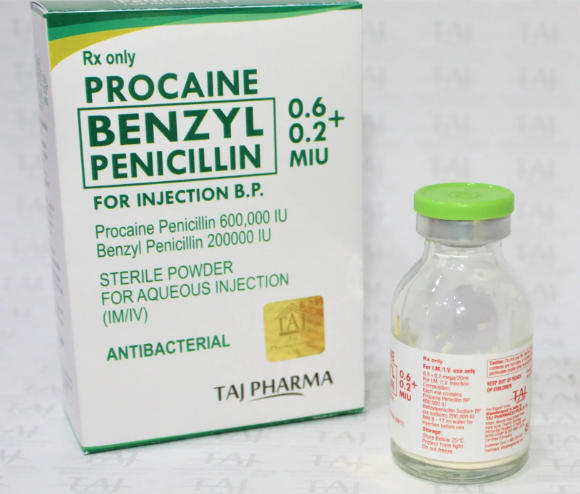
Procaine Penicillin Injection is used for a range of bacterial infections including skin and soft tissue infections, respiratory infections, erysipelas, scarlet fever, and prophylaxis for rheumatic fever, with decisions guided by susceptibility data and clinical judgment.
Administration is intramuscular and performed by trained healthcare professionals in clinical settings, typically into the gluteal or thigh muscles using sterile technique. Dosing frequency depends on the infection and patient factors.
Common effects include injection-site reactions and mild systemic symptoms. Severe allergic reactions are rare but can be life-threatening; patients with known penicillin allergies should avoid this medication, and healthcare providers should monitor for anaphylaxis post-injection.
The procaine component provides local anesthesia to reduce pain upon injection and creates a depot-release effect, extending the duration of penicillin G activity for sustained antibacterial action.
The OEM process involves product customization, packaging options, regulatory documentation, and international shipping. Reach out to the sales team to discuss specifications, certifications (GMP/ISO), lead times, and supportive regulatory services.
[1](https://medlineplus.gov/druginfo/meds/a615052.html)
[2](https://medicalguidelines.msf.org/en/viewport/EssDr/english/benzylpenicillin-penicillin-g-injectable-16682499.html)
[3](https://labeling.pfizer.com/showlabeling.aspx?id=718)
[4](https://www.mayoclinic.org/drugs-supplements/penicillin-g-benzathine-and-penicillin-g-procaine-intramuscular-route/description/drg-20452194)
[5](https://reference.medscape.com/drug/penicillin-g-procaine-999572)
[6](https://clinicaltrials.eu/inn/benzylpenicillin-procaine/)
[7](https://www.youtube.com/watch?v=uEoXv0V-Wyo)
Hot tags: Procaine Penicillin Injection, Antibiotic Treatment, Bacterial Infections, Injection Side Effects, Penicillin G Procaine, Medical Uses, Dosage Information, Injection Administration, Procaine Anesthetic, Healthcare Provider Guidance