Content Menu
● Chemical Source and Composition
>> Industrial Synthesis Methods
>> Key Raw Materials and Reaction Details
>>> Example: Catalytic Hydrogenation
● Quality Control and Manufacturing Standards
● Regulatory Considerations and Compliance
● Pharmaceutical and Chemical Applications
● Market Context: Why Synthetic Sourcing Matters
● OEM and Contract Manufacturing Services
● Frequently Asked Questions (FAQ)
>> 1. What chemicals are used to synthesize procaine?
>> 2. Is procaine found in nature?
>> 3. What makes synthetic procaine preferable over natural products?
>> 4. Can procaine be used as a research chemical?
>> 5. How do I order procaine from a Chinese OEM supplier?
Procaine is a synthetic amino ester-type local anesthetic, first introduced in the early 20th century as a revolutionary alternative to cocaine in medical anesthesia. Today, it is a staple pharmaceutical compound in minor surgery, dentistry, and several clinical applications. For manufacturers, R&D laboratories, and OEM providers seeking to supply or source this compound, understanding its origins—from laboratory synthesis to final pharmaceutical grade—is crucial.[3][7][9]
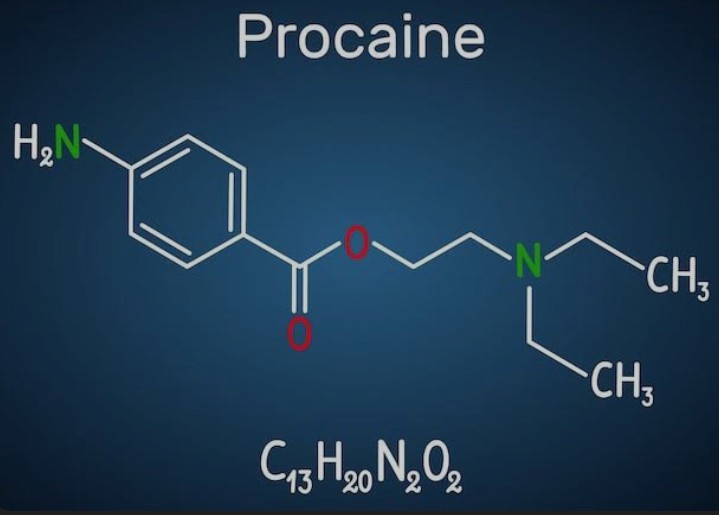
Procaine is not extracted from nature or any organic organism. Its true source lies in the sophisticated processes of synthetic organic chemistry. The active pharmaceutical ingredient is the 2-diethylaminoethyl ester of 4-aminobenzoic acid, more widely known as para-aminobenzoic acid or PABA. These raw materials, sourced from chemical supply chains, are combined under tightly controlled conditions to create the finished drug.[1][8]
There are two main synthetic routes used in laboratories and pharmaceutical factories:
- Direct esterification: Here, ethyl 4-aminobenzoate (benzocaine) reacts with 2-diethylaminoethanol in the presence of sodium ethoxide, typically in an ethanol solution. This pathway is preferred for its high yield and purity.[2][3]
- Oxidative route via nitrobenzoic acid: In this more complex approach, 4-nitrotoluene is oxidized to 4-nitrobenzoic acid, esterified with 2-diethylaminoethanol (often in xylene or ethanol), and the intermediate is then catalytically hydrogenated—most commonly over Raney nickel catalyst and under hydrogen gas pressure—to convert the nitro group to the required amino group.[5][6]
- 4-Aminobenzoic acid (PABA): Typically manufactured through large-scale chemical synthesis. While reminiscent of compounds found naturally in folic acid pathways, commercial sources are always synthetic for quality assurance and purity.[7]
- 2-Diethylaminoethanol: Another industrial intermediate, produced by alkylation and reduction reactions involving diethylamine and ethylene oxide.
- Catalysts and reagents: Sodium ethoxide, thionyl chloride, Raney nickel, hydrogen, ethanol, and various solvents are used in multi-step procedures to yield pharmaceutical-grade procaine.[6][2][5]
In one industrial process, p-nitrobenzoic acid and 2-diethylaminoethanol are esterified to form nitrocaine. This intermediate is then subjected to hydrogenation under high pressure and moderate temperature in the presence of Raney nickel, yielding solid procaine with high efficiency. The process minimizes waste and environmental contamination, which benefits large-scale manufacturers seeking sustainable chemical pathways. Subsequent filtration, extraction, and recrystallization yield the pure active ingredient used in final formulations.[6]
Once synthesized, procaine is typically converted to its hydrochloride salt for use in injection and medical applications. Pharmaceutical-grade manufacturing must meet stringent USP and BP requirements—relying on thorough purity checks using chromatography, spectroscopy, and advanced analytical techniques.[7]
- Sterility: Final product sterility is critical in injectable medications. The conversion to hydrochloride increases water solubility, facilitating safe and effective clinical dosing.[7]
- Consistency: Chemical synthesis offers the repeatability and reliability required to maintain batch-to-batch consistency, especially when targeting the international market.
Before procaine is made available to clients and patients, manufacturers must ensure:
- Full traceability of raw materials
- Compliance with pharmacopoeia standards
- Appropriate handling and documentation for regulatory agencies
International pharmaceutical trade is governed by strict controls on anesthetic agents, and only certified factories with GMP (Good Manufacturing Practice) and ISO accreditations are permitted to export bulk quantities to global customers.

Beyond its central role in anesthesia, procaine:
- Serves as a pharmaceutical intermediate in more complex drug syntheses[7]
- Is investigated as a DNA methylation inhibitor in oncology and epigenetics research[7]
- Functions as a chemical reagent in laboratories studying ion channel blockers and ester chemistry.[7]
As demand grows in research and clinical markets, the ability to synthesize and supply high-purity procaine is a key differentiator for OEM providers.
- Purity and safety: Synthetic procaine is free from biological contaminants, enabling safe use for injections, infusions, and topical applications.
- Supply stability: Chemical synthesis supports scalable production, minimizing the risk of supply chain disruptions.
- Global compliance: Controlled synthetic pathways fulfill the requirements of international regulatory bodies, promoting cross-border trade and OEM partnerships.
At supplybenzocaine.co.uk, our OEM and contract manufacturing services encompass:
- Custom synthesis tailored to client specifications
- Comprehensive R&D support for new formulations, delivery systems, and packaging
- Quality-centric production for international brands, wholesalers, and importers
All processes are designed to meet or exceed the strictest international standards, ensuring our clients receive pharmaceutical-grade procaine suitable for medical, industrial, or research use.
Procaine and its intermediates must be handled with care:
- Storage in controlled, cool, dry environments
- Use of protective equipment during synthesis and handling
- Strictly limited access for unlicensed personnel
Final products are subject to batch testing, serialization, and documentation, further ensuring safety and traceability in global supply networks.
Procaine stands as a testament to the precision and effectiveness of modern synthetic chemistry. Unlike plant-derived pharmaceuticals, its source is entirely industrial, composed of carefully controlled chemical intermediates and advanced processing methods. OEM manufacturers and brand owners seeking consistent, high-quality anesthetic supply can rely on Chinese factories with integrated R&D and production capabilities for safe, compliant, and innovative solutions. For custom orders, process support, and pharmaceutical partnership, contact supplybenzocaine.co.uk—a proven provider for international pharmaceutical markets.

Procaine is synthesized from para-aminobenzoic acid (PABA) and 2-diethylaminoethanol, often using catalysts such as Raney nickel, sodium ethoxide, and solvents like ethanol or xylene.[5][6]
No, procaine is not a natural product. It is created exclusively by chemical synthesis in laboratories and factories.[3][7]
Synthetic routes ensure purity, reproducibility, and safety, while avoiding contaminants and inconsistencies common with natural extracts.[7]
Yes, it is a valuable intermediate and reagent in pharmaceutical, biochemical, and epigenetics research.[7]
Contact a licensed manufacturer such as supplybenzocaine.co.uk with your specifications; ensure compliance with all regulatory requirements for pharmaceutical import and use.
[1](https://pubchem.ncbi.nlm.nih.gov/compound/Procaine)
[2](https://www.sciencedirect.com/topics/pharmacology-toxicology-and-pharmaceutical-science/procaine)
[3](https://en.wikipedia.org/wiki/Procaine)
[4](https://www.youtube.com/watch?v=1x1Yhy0eywg)
[5](https://www.chemicalbook.com/synthesis/procaine.htm)
[6](https://pt.scribd.com/document/395035796/CN1562961A-Method-for-Preparing-Procaine-Google-Patents)
[7](https://www.nbinno.com/article/pharmaceutical-intermediates/chemistry-of-procaine-hydrochloride-synthesis-and-uses)
[8](https://go.drugbank.com/drugs/DB00721)
[9](https://www.sciencedirect.com/topics/chemistry/procaine)
Hot tags: Procaine Source, Procaine History, Procaine Chemical Structure, Procaine Uses, Procaine Synthesis, Procaine Anesthetic, Procaine Side Effects, Procaine Mechanism, Procaine vs Cocaine, Procaine Applications