Content Menu
● What Is Injectable Lidocaine?
● Is It Legal To Buy Injectable Lidocaine?
● Where Clinics And Hospitals Buy Injectable Lidocaine
● Where Distributors And Wholesalers Buy Injectable Lidocaine
● Where OEM Brands And Manufacturers Source Lidocaine
● Why Work With A Chinese OEM Factory For Lidocaine?
● How supplybenzocaine.co.uk Supports Injectable Lidocaine Projects
● Visual Overview: Injectable Lidocaine Supply Chain
● Practical Steps To Buy Injectable Lidocaine As A Business
● Safety And Regulatory Considerations
● How To Use Images And Videos In Your Article
● FAQ
>> 1. What is the best place to buy injectable lidocaine for a clinic?
>> 2. Can individuals buy injectable lidocaine online?
>> 3. How can brands use Chinese OEM factories for injectable lidocaine?
>> 4. What documents should I request from an injectable lidocaine supplier?
>> 5. Does supplybenzocaine.co.uk provide OEM services for lidocaine products?
Injectable lidocaine is a tightly regulated local anesthetic, and the right purchasing channel depends on who you are: a hospital, private clinic, distributor, or OEM brand owner. This comprehensive guide explains legal sources, purchasing strategies, and how professional factories in China (such as supplybenzocaine.co.uk) support global OEM projects for injectable lidocaine and lidocaine-based products.
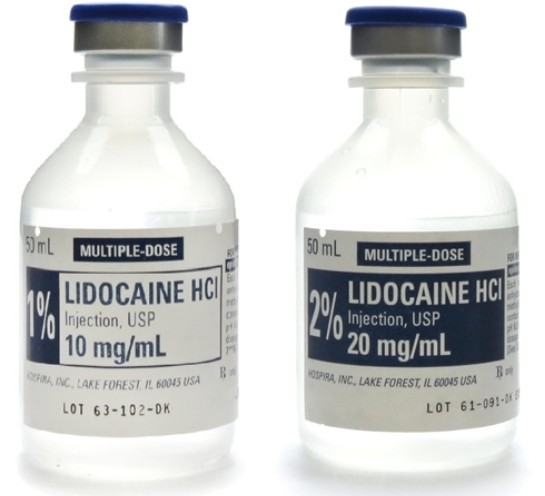
Injectable lidocaine is a local anesthetic used to block nerve conduction and provide temporary numbness in targeted tissues. It is widely used in dentistry, minor surgery, dermatology, cosmetic procedures, emergency medicine, and pain management. By blocking sodium channels in nerve cell membranes, lidocaine prevents the generation and transmission of nerve impulses, resulting in localized loss of sensation.
Most injectable formulations are lidocaine hydrochloride solutions in sterile water, commonly supplied in concentrations such as 0.5%, 1%, or 2%. Some products are combined with epinephrine (adrenaline) to reduce local bleeding and prolong the anesthetic effect by causing vasoconstriction. These solutions are typically filled into ampoules or vials in volumes ranging from 1 mL single-dose units to 50 mL multi-dose containers.
In clinical practice, injectable lidocaine can be administered intradermally, subcutaneously, intramuscularly, intravenously (in controlled indications), or as a nerve block. The route of administration, concentration, and dose depend on the procedure, patient factors, and local regulatory guidelines. Because incorrect dosing or technique can lead to systemic toxicity, cardiac effects, or neurologic complications, handling and use are restricted to trained professionals.
The legality of purchasing injectable lidocaine depends on national and regional regulations. In most major regulatory systems (such as the United States, European Union, Canada, Australia, and China), injectable lidocaine is classified as a prescription medicine intended for use by licensed healthcare professionals. This means that:
- Private individuals generally cannot legally purchase injectable lidocaine for self-use.
- Clinics, hospitals, and licensed practitioners must obtain injectable lidocaine through approved medical supply channels.
- Pharmacy-only or over-the-counter lidocaine products are usually limited to topical formulations (creams, gels, sprays, patches) with lower concentrations for minor pain relief.
For businesses, additional layers of regulation apply. Distributors, wholesalers, and OEM brands must comply with pharmaceutical licensing, pharmacovigilance obligations, controlled storage and transport conditions, and product registration requirements in every country where they sell or distribute injectable lidocaine. Manufacturers and OEM partners must hold appropriate manufacturing licenses, comply with GMP (Good Manufacturing Practice), and provide technical documentation for regulatory submissions.
In short, injectable lidocaine is not a casual consumer product. End users must be qualified professionals, and business buyers must ensure that purchasing, importing, and marketing activities meet all relevant legal and regulatory standards.
Hospitals and clinics usually follow strict procurement pathways to ensure the quality, safety, and traceability of injectable products such as lidocaine. Typical channels include:
- Hospital and institutional pharmacies
Large hospitals often have central pharmacies that manage formulary decisions, supplier contracts, and inventory. Clinicians submit requests, and the pharmacy sources injectable lidocaine from approved pharmaceutical manufacturers or national distributors.
- Authorized medical distributors
Licensed distributors specialize in supplying pharmaceuticals and medical devices to hospitals, clinics, ambulatory surgery centers, dental practices, and emergency services. They typically offer a range of lidocaine injections in standard concentrations (for example, 1% or 2%) and vial sizes.
- Compounding and specialty pharmacies
In cases where clinicians require customized strengths, buffered solutions, or specific combinations (such as lidocaine plus epinephrine within a defined ratio), compounding pharmacies can prepare tailored formulations under pharmacy and medical regulations. This is particularly relevant in pain clinics, dental practices, and specialty outpatient centers.
These channels ensure that the products are stored within recommended temperature ranges, tracked by batch and lot number, and accompanied by proper documentation. They also provide recall capability, which is essential in the rare event of quality issues or safety alerts.
Medical distributors and wholesalers serve as a bridge between manufacturers and healthcare providers. When sourcing injectable lidocaine, they typically work with:
- Multinational pharmaceutical companies
Global pharmaceutical manufacturers produce finished injectable lidocaine according to stringent regulatory requirements. Distributors enter into commercial agreements, secure marketing authorizations where needed, and then supply hospitals and clinics in their territories.
- Regional GMP-certified plants
In markets with local manufacturing capacity, regional plants may produce injectable lidocaine under license or as independent manufacturers. Distributors can obtain exclusive or semi-exclusive rights for distribution in certain regions or market segments.
- Chinese and other international OEM manufacturers
Many distributors choose to partner with OEM factories in China that specialize in local anesthetics. These factories may provide finished vials and ampoules under private labels, or supply bulk solution and packaging services for brands that control distribution.
Key factors that wholesalers and distributors consider include price, quality, regulatory status, continuity of supply, and technical support. Chinese OEM partners often attract distributors with competitive pricing, flexible minimum order quantities, and the ability to customize packaging and branding.
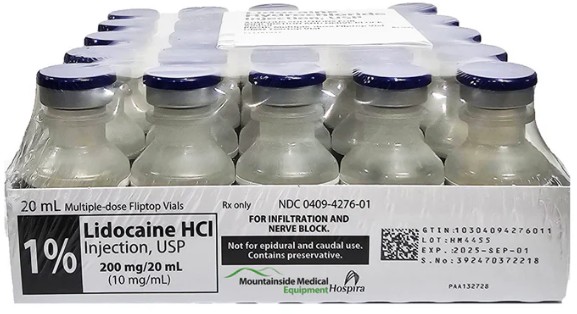
OEM (Original Equipment Manufacturer) brands and pharmaceutical or cosmetic manufacturers often operate at two levels of the supply chain:
1. Active Pharmaceutical Ingredient (API)
Brands or contract manufacturers purchase lidocaine base or lidocaine hydrochloride powder from GMP-compliant API producers. This is the core active ingredient that will later be formulated into injectable or topical products. China is one of the major global hubs for lidocaine API production and export, with established facilities that can supply API at various purity grades and volumes.
2. Finished Injectable or Pre-Formulated Solutions
Some OEM projects involve purchasing bulk sterile lidocaine solution or fully finished injectable products from a factory that can handle formulation, filtration, filling, sterilization, and packaging. The OEM partner may then label the product under the client's brand, meeting the regulatory and language requirements of the target market.
Chinese OEM factories are particularly attractive to brands and manufacturers because they can integrate both levels—API and finished dosage forms—into a one-stop service. This reduces coordination complexity, shortens project timelines, and allows tighter control of costs and quality.
Professional buyers increasingly choose Chinese OEM factories to support their lidocaine portfolios for several reasons:
- Cost-effectiveness
Due to mature supply chains, economies of scale, and efficient production processes, Chinese factories can often deliver lidocaine API and finished products at a significantly lower cost than many local manufacturers, without compromising on quality when GMP standards are consistently applied.
- Scale and flexibility
Many factories are equipped to handle projects ranging from small pilot batches to large-scale commercial production. OEM partners can start with small runs to test new markets or formulations and then scale up once market demand is confirmed.
- Customization and branding
Chinese OEM manufacturers typically offer a wide range of customization: concentration of lidocaine, choice of excipients, vial or ampoule size, type of closure, labeling design, multi-language inserts, and packaging configurations (e.g., hospital bulk packs, clinic cartons, or procedure kits).
- Technical and regulatory support
Experienced OEM partners provide documentation such as Certificates of Analysis, stability data, manufacturing process descriptions, and quality manuals. For many brands, this support is critical when preparing regulatory dossiers and registrations in their home countries.
- Stable supply
A reliable OEM partner can help mitigate supply disruptions by planning production schedules, maintaining safety stocks of key materials, and coordinating logistics to reach multiple countries efficiently.
supplybenzocaine.co.uk is a Chinese factory engaged in the research, development, production, and sales of products in biotechnology, pharmaceutical health, and medical-related fields. For international clients in medical, cosmetic, and related industries, the factory positions itself as a long-term OEM partner rather than a simple trading company.
For lidocaine and similar local anesthetics, the factory can typically offer:
- High-purity lidocaine powder and intermediates
Suitable for injectable formulations and other dosage forms, with specification control according to pharmacopeial or customer-defined standards.
- Technical guidance for formulation
Professional technical staff can offer advice on solution preparation, pH adjustment, buffering options, and process optimization related to numbing injections and other lidocaine products. While each client remains responsible for local regulatory compliance, such support can reduce R&D time and risk.
- OEM and custom manufacturing services
The factory can provide customized services that include formulation (within the regulatory and technical scope), sterile filling of vials or ampoules, packaging and labeling with the client's brand, and batch documentation. Whether you are a brand owner, a wholesaler, or a producer looking to expand your product line, OEM solutions can help you enter or strengthen your position in the anesthetic market.
- Flexible cooperation models
Depending on the client's capabilities, supplybenzocaine.co.uk can either supply only raw materials (such as lidocaine powder), cooperate on semi-finished products, or deliver turnkey finished goods ready for final market distribution.
By combining production strength with international trade experience, the factory aims to simplify cooperation for overseas partners, from initial inquiry and sample evaluation to long-term batch supply.
Although this text does not embed actual images or video links, you can enrich your website article with visual content that closely follows the real-world supply chain of injectable lidocaine. A typical supply chain moves through the following stages:
1. Raw materials and intermediates
Chemical starting materials and intermediates used to synthesize lidocaine are sourced and tested.
2. Lidocaine API production
The API is produced in GMP facilities, where synthesis, purification, and drying are performed. Analytical tests confirm identity, purity, and potency.
3. Formulation and filling
Lidocaine API is dissolved and formulated into sterile solution. The solution is filtered, filled into vials or ampoules, and sealed in controlled cleanroom environments.
4. Sterilization and quality control
Products undergo sterilization (if not terminally sterilized in the filling process), and batches are subjected to microbiological, chemical, and physical tests.
5. Packaging and labeling
Finished injectable units are labeled with dosage, batch number, expiration date, barcodes, and regulatory information, then packed into cartons suitable for hospitals, clinics, or distributors.
6. Distribution and logistics
Products move through distributors and wholesalers to end-users such as hospitals, clinics, and licensed practitioners, often under controlled temperature conditions.
For each of these stages, you can use custom photos, diagrams, or short videos to demonstrate your capabilities or help customers visualize the journey from raw materials to ready-to-use injectable products.
If you are a business buyer rather than an individual, it is useful to approach purchasing injectable lidocaine systematically. The following steps provide a practical roadmap for clinics, distributors, and OEM brands:
1. Define your role and objectives
Clarify whether you are a clinic, distributor, brand owner, or manufacturer. Your position in the supply chain will determine whether you need small professional-use quantities, wholesale volumes, or OEM manufacturing services.
2. Check regulatory requirements in your market
Investigate how injectable lidocaine is classified in your country and what approvals are required. You may need marketing authorization, import licenses, or registration of your brand name and packaging.
3. Decide what you actually need to buy
- Clinics: finished injectable vials from local distributors or institutional pharmacies.
- Distributors: finished injectable products from manufacturers or OEM partners.
- OEM brands: API, bulk solution, or fully finished OEM-labeled products.
4. Evaluate potential suppliers
Shortlist suppliers based on their manufacturing scale, quality systems, pricing, flexibility, and experience with international customers. For Chinese factories, confirm their export experience and familiarity with your destination market's rules.
5. Verify qualifications and documentation
Request GMP or equivalent certifications, quality manuals, sample Certificates of Analysis, and stability data. For OEM projects, also request technical dossiers and packaging templates.
6. Request samples and pilot batches
Before committing to large orders, test samples in-house or through your contracted laboratories. Assess appearance, pH, concentration, sterility, and overall performance in intended applications.
7. Define OEM specifications and sign agreements
Once you are satisfied with quality and technical capabilities, agree on product specs: concentration, container type, labeling design, languages, and packaging unit sizes. Negotiate pricing, minimum order quantities, production lead times, and delivery terms.
8. Plan long-term cooperation and quality monitoring
Establish a quality agreement that defines responsibilities for both parties. Arrange regular reviews, audits where possible, and consistent communication. This helps ensure that quality remains stable even as demand grows.
By following these steps, businesses can reduce risk, improve predictability, and build long-term, mutually beneficial relationships with manufacturers or OEM partners.
Safety must be at the center of any decisions involving injectable lidocaine. Key considerations include:
- Professional administration only
Injectable lidocaine is intended for use by trained healthcare professionals who understand dosing, contraindications, interactions, and emergency management of adverse reactions. It is not suitable for self-injection by untrained individuals.
- Patient screening and dosing
Appropriate dosing depends on body weight, the site of injection, concomitant medications, and coexisting conditions such as heart disease or hepatic impairment. Professional guidelines help clinicians determine maximum safe doses and appropriate combinations with other anesthetics or solutions.
- Product quality and sterility
Since injectable products bypass the body's normal barriers, sterility and particulate control are critical. Quality failures can result in infections, inflammatory reactions, or systemic toxicity.
- Regulatory compliance
Manufacturers, distributors, and OEM brands must comply with pharmaceutical laws, GMP requirements, labeling rules, and pharmacovigilance expectations. Failure to meet these obligations can lead to product recalls, regulatory actions, or legal liabilities.
- Clear labeling and information
Accurate and clear labeling on vials, cartons, and information leaflets helps professionals use products correctly. This includes concentration, route of administration, warnings, expiration dates, storage conditions, and manufacturer details.
By carefully selecting reputable suppliers and ensuring compliance at every stage, businesses can protect patients, professionals, and their own brand reputation.
If you are preparing content for your own website, especially targeted at international buyers or professional partners, you can significantly enhance credibility and engagement with well-chosen images and videos. Consider including:
- Photos of cleanrooms, production lines, and quality-control laboratories to demonstrate your manufacturing strength.
- Close-up images of vial or ampoule packaging that highlight clarity of labeling, batch numbers, and professional presentation.
- Diagrams of the lidocaine supply chain or simplified process flow charts that show how raw materials become injectable products.
- Short factory tour videos that walk potential customers through the receiving area, synthesis or formulation rooms, filtration units, filling machines, and final packaging.
- Professional explainer videos that outline how OEM cooperation works—from initial inquiry and technical discussion to sample verification, contract signing, and shipping.
When you integrate such visuals into your article, place them near relevant sections (for example, manufacturing images in OEM sections and product shots in procurement sections). This helps readers quickly connect the text with real-world capabilities and builds trust in your factory or brand.
Injectable lidocaine is a vital tool in modern medicine and cosmetic practice, but it is tightly regulated and must be handled with professional care. For individuals, the only legitimate access to injectable lidocaine is through licensed healthcare providers who prescribe and administer it safely. For clinics, hospitals, distributors, and OEM brands, choosing the right supply route—whether through local distributors or direct cooperation with international manufacturers—is essential for ensuring long-term, compliant, and cost-effective access.
Chinese OEM factories, such as the team behind supplybenzocaine.co.uk, can play a key role in global supply strategies by offering cost-effective lidocaine API, high-quality formulation and filling services, and flexible branding solutions. With proper regulatory planning and careful supplier selection, foreign brand owners, wholesalers, and producers can build or expand their anesthetic portfolios while maintaining safety and quality standards.
If you are looking for reliable support for lidocaine-related products—whether you need raw materials, bulk injections, or fully customized OEM solutions—you are welcome to contact supplybenzocaine.co.uk. Our team can discuss your market needs, help you evaluate feasible specifications, and work with you step by step from initial samples to stable, long-term supply. Take the next step now and reach out to explore how we can support your injectable lidocaine projects and broader medical product strategy. Contact us to get more information!
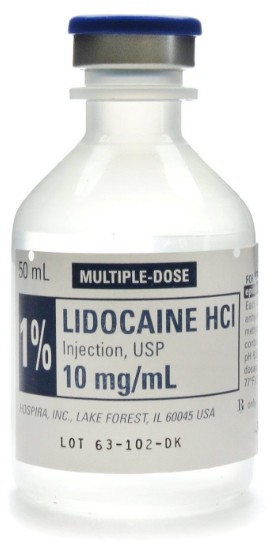
The best place for a clinic or hospital to buy injectable lidocaine is through institutional pharmacies or authorized medical distributors that are approved in the clinic's jurisdiction. These channels ensure that the products are properly stored, documented, and traceable and that they comply with local regulatory requirements. For specialized needs, clinics may also work with licensed compounding pharmacies that can prepare custom formulations under professional supervision.
In most regulated markets, individuals cannot legally buy injectable lidocaine online for self-injection. Injectable formulations are prescription-only and intended for use by trained medical professionals. While some websites may offer such products directly to consumers, purchasing from these sources can be illegal and unsafe, and the quality or authenticity of the product cannot be guaranteed. Individuals who need anesthesia should consult a licensed healthcare provider rather than attempting to self-source injectable lidocaine.
Brands can collaborate with Chinese OEM factories by defining their target market, required concentrations, packaging formats, and regulatory needs, then working with the factory to create custom products. Typically, the brand will request technical documentation, evaluate samples, and conduct stability or performance testing. Once satisfied, the parties sign OEM agreements covering specifications, quality standards, and pricing. The factory then manufactures and packages the injectable lidocaine under the brand's label, while the brand handles local registration, marketing, and distribution.
From an injectable lidocaine supplier, you should request GMP or equivalent manufacturing certifications, Certificates of Analysis for representative batches, stability data, and detailed information on the formulation and manufacturing process. For OEM projects, it is also important to obtain technical dossiers, packaging and labeling mockups, and quality system documentation. These materials help you prepare regulatory submissions, demonstrate compliance to authorities, and evaluate the supplier's long-term reliability.
Yes, supplybenzocaine.co.uk provides OEM services related to lidocaine and other local anesthetics. As a Chinese factory engaged in biotechnology, pharmaceutical health, and medical-related fields, the company can supply lidocaine powder, support formulation work for numbing injections and other products, and offer customized filling, packaging, and branding services for foreign clients. Whether you are a brand owner, wholesaler, or manufacturer, you can work with the factory to develop tailored solutions that meet the requirements of your target market.
1. https://www.olympiapharmacy.com/product/lidocaine-hcl-1-injection/
2. https://www.made-in-china.com/products-search/hot-china-products/Lidocaine_Injection_Factory.html
3. https://www.supplybenzocaine.co.uk/where-can-you-buy-lidocaine.html
4. https://estore.healthfirst.com/product/1-lidocaine-hcl-injection-usp-500mg-50ml-10mg-ml-50ml-vial
5. https://pharmaoffer.com/api-excipient-supplier/local-anesthetics/lidocaine/china
6. https://www.supplybenzocaine.co.uk/how-to-reconstitute-lidocaine-powder-for-numbing-injection.html
7. https://pemcincinnati.com/blog/which-injectable-local-anesthetic-should-i-use/
8. https://www.made-in-china.com/products-search/hot-china-products/Factory_Supply_Lidocaine.html
9. https://www.supplybenzocaine.co.uk/can-you-buy-novocaine.html
10. https://www.boundtree.com/pharmaceuticals/rx-pharmaceuticals/lidocaine-1-10mg-ml-30ml-vial/p/4279-02
Hot tags: Buy Injectable Lidocaine Online, Lidocaine HCl 2% Injection For Sale, Lidocaine Hydrochloride Injection USP, Where To Buy Lidocaine Vials, Lidocaine With Epinephrine Injection, Medical Grade Lidocaine Injection, Dental Local Anesthetic Supplies, Wholesale Injectable Lidocaine, Lidocaine 1% Injection Price, Sterile Lidocaine Vials Online